Spectinomycin
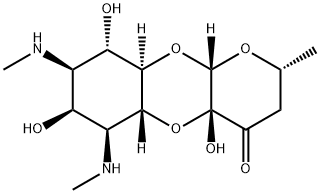
- CAS No.
- 1695-77-8
- Chemical Name:
- Spectinomycin
- Synonyms
- espectinomicina;trobicin;spectinomycine;spectam;togamycin;Spectacin;macromycin;pectinomycin;SPECTINOMYCIN;spectinomicina
- CBNumber:
- CB8197632
- Molecular Formula:
- C14H24N2O7
- Molecular Weight:
- 332.35
- MOL File:
- 1695-77-8.mol
- MSDS File:
- SDS
- Modify Date:
- 2022/12/21 16:56:50
| Melting point | 185 °C (decomp) |
|---|---|
| Boiling point | 583.1±50.0 °C(Predicted) |
| alpha | D25 -20° (water) |
| Density | 1.43±0.1 g/cm3(Predicted) |
| pka | pKa1 6.95, pKa2 8.70(at 25℃) |
| EPA Substance Registry System | Spectinomycin (1695-77-8) |
Spectinomycin Chemical Properties,Uses,Production
Description
An unusual aminoglycoside antibiotic, spectinomycin is produced by fermentation of Streptomyces spectabil is and differs substantially in its clinical properties from the others. The diaminoinositol unit (spectinamine) contains two mono-N-methyl groups, and the hydroxyl between them has a stereochemistry opposite to that in streptomycin. The glycosidically attached sugar also is unusual in that it contains three consecutive carbonyl groups, either overt or masked, and is fused by two adjacent linkages to spectinamine to produce an unusual, fused, three-ring structure.
Uses
Spectinomycin finds principal application in the treatment of gonorrhea. It should be noted that the antibiotic resistance among N. gonorrhoeae has caused a number of therapeutic problems. It has been found that only by escalating the antibiotic doses and using probenecid to retard the excretion of penicillin and ampicillin (the drugs of choice) has the continued effective use of penicillin, ampicillin, and tetracycline been possible. Even with modifications in the therapy, from 3 to 8% of cases fail to respond to the usual regimens for uncomplicated gonorrhea. Thus, the treatment of uncomplicated gonorrhea that fails to respond to the usual regimen is spectinomycin therapy.
Definition
ChEBI: An organic heterotricyclic antibiotic that is active against gram-negative bacteria and used (as its dihydrochloride pentahydrate) to treat gonorrhea. It is produced by the bacterium Streptomyces spectabilis.
Antimicrobial activity
Its activity is modest and markedly
affected by medium composition and pH. It exerts only moderate
activity against Gram-positive organisms. It is widely
active against enterobacteria, but Providencia spp. are resistant.
Anaerobic bacteria are also resistant.
Of particular interest is its activity against N. gonorrhoeae,
including β-lactamase-producing strains. Among other sexually
acquired organisms, Ureaplasma urealyticum is susceptible,
but Chlamydia trachomatis and T. pallidum are resistant.
For most organisms, the minimum bactericidal concentration
(MBC) is at least four times the MIC and it is regarded
as essentially bacteristatic. In contrast, it is bactericidal for
gonococci at concentrations close to the MIC, which is of the
order of 2–16 mg/L for both penicillin-susceptible and resistant
strains.
Acquired resistance
N. gonorrhoeae strains resistant to spectinomycin have emerged
in South East Asia, the USA and the UK; the resistance of
UK isolates was not attributable to aminoglycoside-modifying
enzymes. In most countries where its use remains low the
prevalence of resistance in gonorrhea is also low.
Acquired resistance in enterobacteria, enterococci and staphylococci
can be caused by nucleotidyltransferases that modify
the drug at position 9. The enzyme from Gram-negative organisms
ANT(3″) (9) also modifies streptomycin at position 3″,
thus conferring cross-resistance to the two drugs. There is no
enzymatic cross-resistance with 2-deoxystreptamine-
containing
aminoglycosides.
Contact allergens
Spectinomycin is an aminocyclitol antibiotic. It is used in human medicine against Neisseria gonorrhoeae and in veterinary medicine, especially for poultry, pigs, and cattle. Cases of dermatitis have been reported in veterinary practice.
Pharmacokinetics
Cmax 25 mg/kg intramuscular: 60–80 mg/L after 1 h
Plasma half-life: 2–3 h
Volume of distribution: 10–13.4 L
Plasma protein binding: <10%
It is poorly absorbed on oral administration. It is almost completely
excreted unchanged in the urine over 48 h, concentrations
on conventional dosage reaching 1 g/L. Excretion is prolonged in renal impairment, and is unaffected by
probenecid.
Clinical Use
Gonorrhea in penicillin-allergic patients or due to penicillin-resistant strains (single-dose treatment)
Side effects
Transient headache, dizziness, pain at the site of injection and occasional fever have been described. No evidence of ototoxicity or renal toxicity has been found in volunteers receiving doses of 2 g every 6 h for 3 weeks, amounts much in excess of those used therapeutically.
Spectinomycin Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| career henan chemical co | +86-0371-86658258 +8613203830695 | China | 29826 | 58 | Inquiry |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 | China | 12459 | 58 | Inquiry |
| Hefei TNJ Chemical Industry Co.,Ltd. | 0551-65418671 | China | 34571 | 58 | Inquiry |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-89586680 +86-18192503167 | China | 9030 | 58 | Inquiry |
| Dorne Chemical Technology co. LTD | +86-86-13583358881 +8618560316533 | China | 3148 | 58 | Inquiry |
| AFINE CHEMICALS LIMITED | +86-0571-85134551 | China | 15395 | 58 | Inquiry |
| BOC Sciences | 16314854226; +16314854226 | United States | 19743 | 58 | Inquiry |
| Dayang Chem (Hangzhou) Co.,Ltd. | 571-88938639 +8617705817739 | China | 52861 | 58 | Inquiry |
Related articles
- An aminocyclitol compound:Spectinomycin
- Spectinomycin is an aminocyclitol compound which has some structural similarities to streptomycin, but it differs by not being....
- Mar 22,2022
1695-77-8(Spectinomycin)Related Search:
1of4
chevron_right




