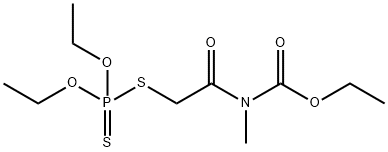
MECARBAM
- CAS No.
- 2595-54-2
- Chemical Name:
- MECARBAM
- Synonyms
- p474;afos;TD 72;mc474;ms1053;ms1143;pestan;muratox;murutox;MURFOTOX
- CBNumber:
- CB9773912
- Molecular Formula:
- C10H20NO5PS2
- Molecular Weight:
- 329.37
- MOL File:
- 2595-54-2.mol
- Modify Date:
- 2023/4/23 13:52:06
| Melting point | <25℃ |
|---|---|
| Boiling point | 144°C |
| Density | 1.222 g/cm3 (20 ºC) |
| vapor pressure | 5×10-5 Pa (20 °C, est.) |
| refractive index | 1.5138 (589.3 nm 20℃) |
| Flash point | >100 °C |
| storage temp. | APPROX 4°C |
| form | liquid |
| Water Solubility | 850 mg l-1 (est .) |
| pka | -2.42±0.70(Predicted) |
| BRN | 1888011 |
| EPA Substance Registry System | Mecarbam (2595-54-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS09 |
|---|---|
| Signal word | Danger |
| Hazard statements | H300+H330-H311-H410 |
| Precautionary statements | P260-P264-P273-P280-P302+P352+P312-P304+P340+P310 |
| Hazard Codes | T,N,Xn,F |
| Risk Statements | 24/25-50/53-36-20/21/22-11 |
| Safety Statements | 36/37-45-60-61-36-26-16 |
| RIDADR | 3018 |
| WGK Germany | 3 |
| RTECS | FB3850000 |
| HazardClass | 6.1(a) |
| PackingGroup | II |
| HS Code | 29309090 |
| Toxicity | LD50 oral in rabbit: 60mg/kg |
MECARBAM price More Price(2)
MECARBAM Chemical Properties,Uses,Production
Description
Mecarbam is a pale yellow to light brown oil, bp 144 ?C/0.02 mm Hg. It is slightly soluble in water (<0.1% at 25 ?C) and miscible with most organic solvents except alkanes. Log Kow = 2.6. It is readily hydrolyzed in aqueous media below pH 3.
Chemical Properties
Yellow oil.
Uses
Mecarbam is an organothiophosphate acaricides used to control powdery mildew of cucumbers.
Definition
ChEBI: An organic thiophosphate that is O,O-diethyl hydrogen phosphorodithioate in which the hydrogen attached to a sulfur is replaced by a 2-[(ethoxycarbonyl)(methyl)amino]-2-oxoethyl group.
Hazard
Highly toxic, cholinesterase inhibitor. Use may be restricted.
Metabolic pathway
Mecarbam in a close chemical analogue of dimethoate and it is photolytically decarboxyethylated to the diethyl analogue of dimethoate. The main route of hydrolytic decomposition in moderately basic solution is via attack on the S-methylene carbon atom rather than on phosphorus or the carbamoyl group, followed by cleavage of the S-C bond yielding O,Odiethyl phosphorodithioate as the main product. Mecarbam oxon has been reported as a metabolite in plants and animals but few detailed studies have been reported on the metabolic or environmental fate of mecarbam.
Metabolism
In mammals, mecarbam is rapidly metabolized by hydrolysis, oxidative desulfuration to the oxon, and degradation of the carbamoyl moiety. O-Deethylation also takes place to a minor extent. In soil, it persists for 4–6 weeks.
MECARBAM Preparation Products And Raw materials
Raw materials
Preparation Products
2595-54-2(MECARBAM)Related Search:
1of3
chevron_right




