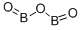삼산화붕소(무수붕산)
|
|
삼산화붕소(무수붕산) 속성
- 녹는점
- 450 °C(lit.)
- 끓는 점
- 1860 °C
- 밀도
- 2.46 g/mL at 25 °C(lit.)
- 증기 밀도
- >1 (vs air)
- 증기압
- 1Pa
- 인화점
- 1860°C
- 저장 조건
- Inert atmosphere,Room Temperature
- 용해도
- 36g/L
- 물리적 상태
- 펠릿
- Specific Gravity
- 2.46 +/- 0.01
- 색상
- 하얀색
- 냄새
- 냄새 없는
- 수소이온지수(pH)
- 4 (10g/l, H2O, 25℃)
- 수용성
- 36g/L(25℃)
- 감도
- Hygroscopic
- Merck
- 14,1337
- 노출 한도
- ACGIH: TWA 10 mg/m3
OSHA: TWA 15 mg/m3
NIOSH: IDLH 2000 mg/m3; TWA 10 mg/m3
- 안정성
- 안정적인. 수분에 민감합니다. 물과 호환되지 않습니다.
- InChIKey
- JKWMSGQKBLHBQQ-UHFFFAOYSA-N
- CAS 데이터베이스
- 1303-86-2(CAS DataBase Reference)
안전
- 위험 및 안전 성명
| 위험품 표기 | Xi,T | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/37/38-61-60 | ||
| 안전지침서 | 26-37/39-45-53 | ||
| OEB | B | ||
| OEL | TWA: 10 mg/m3 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | ED7900000 | ||
| F 고인화성물질 | 3-10 | ||
| TSCA | Yes | ||
| HS 번호 | 28100010 | ||
| 유해 물질 데이터 | 1303-86-2(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: 3150 mg/kg | ||
| IDLA | 2,000 mg/m3 | ||
| 기존화학 물질 | KE-09919 | ||
| 유해화학물질 필터링 | 2012-1-640 | ||
| 중점관리물질 필터링 | 별표1-99 | ||
| 함량 및 규제정보 | 물질구분: 유독물질; 혼합물(제품)함량정보: 삼산화 이붕소 및 이를 0.3% 이상 함유한 혼합물 |
삼산화붕소(무수붕산) C화학적 특성, 용도, 생산
화학적 성질
Boron oxide is a noncombustible, colorless, semitransparent lumps or hard, white, odorless crystals, with slightly bitter taste.
물리적 성질
Colorless glassy solid or vitreous crystal; hexagonal crystal system; slightly bitter taste; hygroscopic; density 2.55 g/cm3; melts at 450°C; vaporizes at 1,500°C; slightly soluble in cold water (3.3%), soluble in alcohol and boiling water (20%).용도
Boron oxide was used as the intermediate glass layer at a bonding temperature of 450°C. In preparation of fluxes; component of enamels and glass; catalyst in organic reaction.In metallurgy; in analysis of silicates to determine SiO2 and alkalies; in blowpipe analysis.
제조 방법
Boric oxide is produced by treating borax with sulfuric acid in a fusion furnace. At temperatures above 750°C, the molten boric acid layer separates out from sodium sulfate. It then is decanted, cooled, and obtained in 96-97% purity. Boric acid above 99% purity may be obtained by fusing granular material.Boric oxide may be prepared by heating boric acid.
2B(OH)3 → B2O3 + 3H2O
생산 방법
Boric oxide is produced by thermal fusion of boric acid, forming a clear transparent glass-like solid that is subsequently ground into white vitreous granules. It is used principally in the manufacture of glass and vitreous products.일반 설명
Colorless, semi-transparent glassy lumps or hard white odorless crystals. Mp 450°C; bp: 1860°C. Density: 2.46 g cm-3. Moderately soluble in water. Used as an insecticide; as the starting material for the synthesis of other boron compounds; as a fluxing agent in enamels and glasses; and in mixture with 2-6% boron nitride, as a bonding agent in the hot isostatic pressing of boron nitride ceramics.반응 프로필
Boron oxide is non-combustible. Of generally low chemical reactivity. Reacts exothermically but slowly with water to form boric acid, a weak acid. Reacts exothermically with strong bases. May react with strong reducing agents such as metal hydrides, metal alkyls to generate flammable or explosive gases. May react violently on contact with bromine pentafluoride. Corrosive to metals in the presence of air.위험도
Eye and upper respiratory tract irritant.건강위험
Boron oxide is an eye and respiratory irritant. In 113 workers exposed to boron oxide and boric acid dusts, there were statistically significant increases in symptoms of eye irrita- tion; dryness of the mouth, nose, and throat; sore throat; and productive cough compared with controls. The mean exposure level was 4.1mg/m3 , with a range of 1.2–8.5mg/m3 . Exposures may occasionally have exceeded 10 mg/m3 . Because of mixed exposures, the study does not indicate whether boron oxide or boric acid dust is more important in causing symp- toms, nor does it indicate the minimum duration of exposure necessary to produce symptoms.Excessive absorption of boron oxide may lead to cardiovascular collapse, alterations in temperature regulation, and coma.
잠재적 노출
Boron oxide is used in glass manufacture and the production of other boron compounds. It is used in fluxes, enamels, drying agents, and as a catalyst.운송 방법
UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9—Miscellaneous hazardous material, Technical Name Required.비 호환성
Incompatible with bromine pentafluoride, calcium oxide. Reacts slowly with water, forming boric acid. Reacts exothermically with alkaline material and strong bases. May react with strong reducing agents such as metal hydrides, metal alkyls to generate flammable or explosive gases. May react violently on contact with bromine pentafluoride. Corrosive to metals in the presence of air.삼산화붕소(무수붕산) 준비 용품 및 원자재
원자재
준비 용품
삼산화붕소(무수붕산) 공급 업체
글로벌( 383)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Anhui Royal Chemical Co., Ltd. | +86-25-86655873 +8613962173137 |
marketing@royal-chem.com | China | 189 | 55 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2503 | 58 |
| Hebei Zhuanglai Chemical Trading Co.,Ltd | +8613343047651 |
admin@zlchemi.com | China | 3002 | 58 |
| Shandong Deshang Chemical Co., Ltd. | +86-0531-8875-2665 +8613153039501 |
info@deshangchem.com | China | 518 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21673 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2989 | 55 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 |
zheyansh@163.com | CHINA | 3620 | 58 |
| QINGDAO HONG JIN CHEMCIAL CO.,LTD. | 532-83657313 |
hjt@hong-jin.com | China | 228 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8827 | 58 |