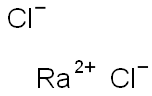염화라듐 C화학적 특성, 용도, 생산
개요
Radium chloride, RaCl2, was the first radium
compound to be prepared in a pure state and was the
basis of Marie Curie’s original separation of radium
from Barium. The first preparation of radium metal
was by the electrolysis of a solution of radium chloride
using a mercury cathode. The structure remains
unknown.
Radium chloride is soluble and crystallizes from solution
as the dihydrate.
RaCl2 is used to produce radon gas that in turn is
used as a cancer treatment in medicine. This compound
is not offered for sale to the general public.
화학적 성질
radium chloride is yellowish-white crystals becoming yel- low or pink on standing. Radiactive. Soluble in water and alcohol.
물리적 성질
radium chloride is a white solid having a blue-green luminescence, especially when heated. It is less soluble in water than other alkaline earth chlorides, a fact which is used in the first stages of the separation of radium from barium by fractional crystallization. It is only sparingly soluble in azeotropic HCl (20.2% HCl) and virtually insoluble in concentrated hydrochloric acid.
용도
Radium chloride (RaCl2) has the same use as radium bromide.
위험도
As for radium.
염화라듐 준비 용품 및 원자재
원자재
준비 용품