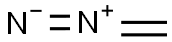다이아조메테인(디아조메탄) C화학적 특성, 용도, 생산
화학적 성질
Diazomethane is a flammable, yellow gas or
a liquid under pressure. Musty odor.
용도
Powerful methylating agent for acidic Compounds such as carboxylic acids, phenols, enols. For syntheses with diazomethane see the reviews by Smith, Chem. Rev. 23, 193 (1938); Eistert, Z. Angew. Chem. 54, 99, 124 (1941) translated by Spangler in Newer Methods of Preparative Organic Chemistry (New York, 1948) p 513; J. S. Pizey, Synthetic Reagents vol. 2 (John Wiley, New York, 1974) pp 65-142.
정의
ChEBI: The simplest diazo compound, in which a diazo group is attached to a methylene group.
일반 설명
Yellow gas with a musty odor. Highly toxic by inhalation Shipped as a liquid under pressure.
공기와 물의 반응
Reacts with water, releasing nitrogen, more stable in ether or dioxane.
반응 프로필
Diazomethane undergoes violent thermal decomposition. Above 200°C. the vapors may explode violently if rough glass surfaces are present. Explosions at low temperatures can occur if traces of organic matter are present. [J. Phys. Chem. 35:1403(1931)]. Produces explosions with alkali metals. Reacts with copper powder and to some extent all solid surfaces to produce nitrogen and solid white polymethylene. Reacts with dimethylaminodimethylarsine and trimethyltin in ether with vigorous foaming.
건강위험
Diazomethane vapor causes severe irritation of the skin, eyes, mucous membranes,
and lungs. It is considered to be a substance with poor warning properties, and the
effects of exposure may be delayed in onset. Symptoms of exposure may include
headache, chest pain, cough, fever, severe asthmatic attacks, and pulmonary edema,
which can be fatal. Exposure of the skin and mucous membranes to diazomethane
may cause serious burns.
Diazomethane is a powerful allergen. Prolonged or repeated exposure to
diazomethane can lead to sensitization of the skin and lungs, in which case asthma-
like symptoms or fever may occur as the result of exposure to concentrations of
diazomethane that previously caused no symptoms. Chronic exposure to
diazomethane has been reported to cause cancer in experimental animals, but this
substance has not been identified as a human carcinogen.
Note that diazomethane is often prepared in situ from precursors that may
themselves be highly toxic and/or carcinogenic.
화재위험
Pure diazomethane gas and liquid are readily flammable and can explode easily. A
variety of conditions have been reported to cause explosions of diazomethane,
including contact with rough surfaces such as ground-glass joints, etched or
scratched flasks, and glass tubing that has not been carefully fire-polished. Direct
sunlight and strong artificial light may also cause explosions of this substance.
Violent reactions may occur on exposure of diazomethane to alkali metals.
인화성 및 폭발성
Pure diazomethane gas and liquid are readily flammable and can explode easily. A variety of conditions have been reported to cause explosions of diazomethane, including contact with rough surfaces such as ground-glass joints, etched or scratched flasks, and glass tubing that has not been carefully fire-polished. Direct sunlight and strong artificial light may also cause explosions of this substance. Violent reactions may occur on exposure of diazomethane to alkali metals.
Safety Profile
Confirmed carcinogen
with experimental tumorigenic data. A
poisonous irritant by inhalation. A powerful
allergen. It can cause pulmonary edema and
frequently causes hypersensitivity leading to
asthmatic symptoms. Mutation data
reported. Highly explosive when shocked,
exposed to heat, or by chemical reaction.
Undiluted liquid or gas may explode on
contact with alkali metals, rough surfaces,
heat (lOO°C), hgh-intensity light, or shock.
When heated to decomposition or on
contact with acid or acid fumes it emits
highly toxic fumes of NOx. Incompatible
with alkali metals; calcium sulfate.
잠재적 노출
Diazomethane is a powerful methylat-
ing agent for acidic compounds, such as carboxylic acids,
phenols and enols. It is used in pesticide manufacture and
pharmaceutical manufacture.
Carcinogenicity
Diazomethane was administered
to rats and mice by inhalation, dermal, or subcutaneous
injection routes using concentrations of 0.1 or 3.3 mg/mL.
Mice developed lung tumors following either dermal application
or inhalation at both concentrations.
저장
diazomethane should preferably be handled in solution using glassware specially designated for diazomethane (e.g., with Clear-Seal joints) and should be used as soon as possible after preparation. Storage of diazomethane solutions (even at low temperature) is not advisable. All work with diazomethane should be conducted in a fume hood behind a safety shield, and appropriate impermeable gloves, protective clothing, and safety goggles should be worn at all times.
운송 방법
UN1953 Compressed gas, toxic, flammable, n.o.s.
비 호환성
Heat (at about or above 100
C), shock,
friction, concussion, sunlight, or other intense illuminations
may cause explosions. Contact with alkali metals; drying
agents such as calcium sulfate, or rough edges (such as
ground glass) may cause explosions. Diazo compounds can
detonate. This applies in particular to organic azides that
have been sensitized by the addition of metal salts or strong
acids. Toxic gases are formed by mixing materials of this
class with acids, aldehydes, amides, carbamates, cyanides,
inorganic fluorides, halogenated organics, isocyanates,
ketones, metals, nitrides, peroxides, phenols, epoxides, acyl
halides, and strong oxidizing or reducing agents.
Flammable gases are formed by mixing materials in this
group with alkali metals. Explosive combination can occur
with strong oxidizing agents, metal salts, peroxides, and
sulfides. This chemical is sensitive to prolonged exposure
to heat. This chemical is incompatible with strong
oxidizing agents
.
폐기물 처리
Decompose chemically
with ceric ammonium nitrate under constant agitation and
cooling
.
주의 사항
Diazomethane is attractive as a methylating agent for carboxylic acids and phenols because it reacts quickly and highly efficiently with the production of only N2 as a by-product (Black, 1983). Its natural yellow color is discharged as it reacts, providing automatic indication of reaction progress. However, because diazomethane is highly toxic, it should be generated and used only in a well-functioning fume hood. Because it explodes on contact with some metals or ground glass of any type (joints, stoppers, syringes, stopcocks), it should be handled behind a safety shield, and other personal protective equipment should be used. Because it has a boiling point of ?23°C, it is usually handled in the ethereal solutions in which it is generated. Because it explodes on contact with CaSO4, its solutions or vapors must never be dried with drierite. Despite all of these hazards, it can be worked with safely, provided that appropriate precautions are observed.
다이아조메테인(디아조메탄) 준비 용품 및 원자재
원자재
준비 용품