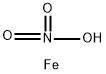
질산철(II). C화학적 특성, 용도, 생산
개요
Iron(II) nitrate is obtained in hydrated form by crystallization from solutions of
iron in cold dilute nitric acid,
4Fe+10HNO3 -> 4Fe(N03)2+NH4N03+3H20
or from the filtrate in the iron(II) sulphate-lead nitrate reaction. At room temperature it
crystallizes as the green hexahydrate; a 9-hydrate is stable below -10°. Solutions of the
nitrate are unstable to heat, nitric oxide being evolved and the iron being oxidized to
iron(III).
화학적 성질
green solid [CRC10]
질산철(II). 준비 용품 및 원자재
원자재
준비 용품