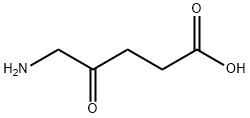
5-아미노레불린산 C화학적 특성, 용도, 생산
개요
5-aminolevulinic acid is the simplest delta-amino acid in which the hydrogens at the gamma position are replaced by an oxo group. It is metabolised to protoporphyrin IX, a photoactive compound which accumulates in the skin. Used (in the form of the hydrochloride salt)in combination with blue light illumination for the treatment of minimally to moderately thick actinic keratosis of the face or scalp. It has a role as a photosensitizing agent, an antineoplastic agent, a dermatologic drug, a prodrug, a plant metabolite, a human metabolite, a Saccharomyces cerevisiae metabolite, an Escherichia coli metabolite and a mouse metabolite. It is a delta-amino acid and a 4-oxo monocarboxylic acid. It derives from a 4-oxopentanoic acid. It is a conjugate base of a 5-ammoniolevulinic acid. It is a conjugate acid of a 5-aminolevulinate. It is a tautomer of a 5-ammoniolevulinate.
용도
5-Aminolevulinic acid (ALA), a nonprotein amino acid involved in tetrapyrrole synthesis, has been widely applied in agriculture, medicine, and food production.
5-Aminolevulinic acid (5-ALA) is an intermediate in heme biosynthesis and is useful in cancer treatment. It is a non-protein amino acid. 5-ALA also has applications in the field of agriculture. It is being studied as an inducing reagent for protoporphyrin IX (PPIX) dependent fluorescence diagnosis of metastatic lymph nodes. 5-ALA is used for photodynamic therapy of diseases, such as Paget′s disease and HPV infection-associated cervical condylomata acuminata.
Intermediate in heme biosynthesis.
Indications
Aminolevulinic acid (ALA HCl, Levulan Kerastick) is
indicated for the treatment of nonhyperkeratotic actinic
keratosis of the face and scalp. It has two components,
an alcohol solution vehicle and ALA HCl as a dry solid.
The two are mixed prior to application to the skin.
When applied to human skin, ALA is metabolized to
protoporphyrin, which accumulates and on exposure to
visible light produces a photodynamic reaction that
generates reactive oxygen species (ROS).The ROS produce
cytotoxic effects that may explain therapeutic efficacy.
Local burning and stinging of treated areas of skin
due to photosensitization can occur.
Biosynthesis
5-ALA was confirmed as the common precursor of tetrapyrrole compounds and was found in many organisms. Two natural 5-ALA biosynthesis pathways are known to date. One is the C4 pathway (Shemin pathway), which Shemin and Russell first reported. It exists in animals, yeast, some protozoa, and purple non-sulfur photosynthetic bacteria. In this pathway, glycine and succinyl-CoA are condensed to 5-ALA under the catalysis of 5-aminolevulinic acid synthase (ALAS), with pyridoxal 5′-phosphate (PLP) as the cofactor. Succinyl-CoA is synthesized by methylmalonyl-CoA mutase, which utilizes vitamin B12 as an essential cofactor (38). The other is the C5 pathway (Beale pathway), which starts from the discovery of 5-ALA in Chlorella vulgaris by Beale. Glutamate produced via the TCA cycle acts as the substrate of the C5 pathway. The C5 pathway is mainly present in higher plants, algae, and many bacteria. The pathway starts with ligating tRNA and glutamate to generate L-glutamyl–tRNA, catalyzed by glutamyl–tRNA synthetase (GluTS). The NADPH-dependent glutamyl-tRNA reductase (GluTR) reduces the carboxyl group of Glu–tRNA to a formyl group, which enables the conversion of L-Glu–tRNA into L-glutamic acid 1-semialdehyde (GSA). In the last step, 5-ALA is created through transamination by glutamate-1-semialdehyde aminotransferase (GSA-AM). The enzymes involved in this pathway, GluTS, GluTR, and GSA-AM, are encoded by gltX, hemA (homonymic with the ALAS gene of the C4 pathway), and hemL, respectively. However, a few microorganisms have C4 and C5 pathways, Euglena gracilis being a well-known example[1].
생물학적 활성
5-Aminolevulinic acid (5-ALA) is a precursor in the biosynthesis of porphyrins, including heme. The conversion of 5-ALA to protoporphyrins within tissues produces a photosensitive target that produces reactive oxygen species upon exposure to light.1 In this way, it is used in photodynamic therapy for a range of dermatological conditions, cancers, and other diseases. Also, oral administration of 5-ALA leads to the preferential accumulation of the fluorescent molecule protoporphyrin IX within certain types of cancer cells. This allows fluorescence-based identification of tumor tissue for accurate resection of diseased tissue.
효소 저해제
This key metabolic precursor (FW = 131.13 g/mol; CAS 106-60-5; pKa values = 4.05 and 8.90 at 25°C; Symbol: ALA), also known as daminolevulinic acid, is essential for the biosynthesis of metal ion-binding tetrapyrrole ring systems (porphyrins, chlorophylls, and cobalamins). In non-photosynthetic eukaryotes (animals, insects, fungi, protozoa, and alphaproteobacteria), d-aminolevulinic acid is produced by the enzyme ALA synthase, using glycine and succinyl CoA as substrates. In plants, algae, bacteria, and archaea, it is produced from glutamyl-tRNA and glutamate-1-semialdehyde. 5-Aminolevulinic acid inhibits (R)-3-amino-2- methylpropionate:pyruvate aminotransferase. ALA Phototherapy: Protoporphyrin IX, the immediate heme precursor is a highly effective tissue photosensitizer that is synthesized in four steps from 5- aminolevulinic acid. ALA synthesis is regulated via a feedback inhibition and gene repression mechanism linked to the concentration of free heme. In certain cell and tissue types, addition of exogenous ALA bypasses these regulation mechanisms, inducing uptake and synthesis of photosensitizing concentrations of Protoporphyrin IX, or PpIX. Topical application of ALA to certain malignant and non-malignant skin lesions, for example, can induce a clinically useful degree of lesion-specific photosensitization (e.g., superficial basal cell carcinomas show high response rate (~79%) after a single phototherapy treatment). ALA also induces localized tissue-specific photosensitization, when injected intradermally. In this sense, ALA and its methyl ester (methyl aminolevulinate, or MAL; trade name: Metvix?) are prodrugs that increase the amounts of the active drug (PpIX).
5-아미노레불린산 준비 용품 및 원자재
원자재
준비 용품