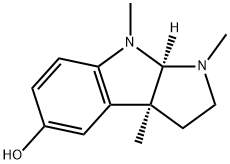
에세롤린
|
|
에세롤린 속성
- 녹는점
- 128 °C
- 끓는 점
- 366.3±42.0 °C(Predicted)
- 밀도
- 1.160±0.06 g/cm3(Predicted)
- 산도 계수 (pKa)
- 10.17±0.40(Predicted)
- InChI
- InChI=1S/C13H18N2O/c1-13-6-7-14(2)12(13)15(3)11-5-4-9(16)8-10(11)13/h4-5,8,12,16H,6-7H2,1-3H3/t12-,13+/m1/s1
- InChIKey
- HKGWQUVGHPDEBZ-OLZOCXBDSA-N
- SMILES
- N1(C)C2=C(C=C(O)C=C2)[C@]2(C)CCN(C)[C@]12[H]
안전
에세롤린 C화학적 특성, 용도, 생산
개요
Eseroline is a drug which acts as an opioid agonist. It is a metabolite of the acetylcholinesterase inhibitor physostigmine but unlike physostigmine, the acetylcholinesterase inhibition produced by eseroline is weak and easily reversible, and it produces fairly potent analgesic effects mediated through the μ-opioid receptor.화학적 성질
Physostigmine crystals or solutions, when heated, exposed to light and air, or in the presence of traces of metal, readily turn red, are more unstable to bases, and are readily hydrolyzed to eserobine, which are capable of being oxidized to red physostigmine salicylate, depriving the product of its therapeutic efficacy.출처
Eseroline, although derived from physostigmine by hydrolysis of N-methylcarbamoil group, is also structurally related to morphine.용도
(-)-Eseroline is a metabolite of physostigmine, displaying both anti-acetylcholinesterase and opiate agonist activities.정의
ChEBI: Eseroline is a pyrroloindole that is 1,2,3,3a,8,8a-hexahydropyrrolo[2,3-b]indole substituted by methyl groups at positions 1, 3a and 8 and a hydroxy group at position 5. It is a metabolite of physostigmine and causes neuronal cell death by a mechanism involving loss of cell ATP. It has a role as an opioid analgesic and a human xenobiotic metabolite. It is a member of phenols and a pyrroloindole.참고 문헌
[1] RENZI G, BARTOLINI A, GALLI A, et al. The structure and action of eseroline: A new antinociceptive drug[J]. Inorganica Chimica Acta, 1980, 40. DOI:10.1016/S0020-1693(00)92145-5.[2] SOMANIS M KrishnaG KuttyR K. Eseroline, a metabolite of physostigmine, induces neuronal cell death.[J]. Toxicology and applied pharmacology, 1990. DOI:10.1016/0041-008x(90)90102-z.
[3] BARTOLINI A, RENZI G, GALLI A, et al. Eseroline: A new antinociceptive agent derived from physostigmine with opiate receptor agonist properties. Experimental in vivo and in vitro studies on cats and rodents[J]. Neuroscience Letters, 1981, 25: 179-183. DOI:10.1016/0304-3940(81)90328-1.
에세롤린 준비 용품 및 원자재
원자재
준비 용품
에세롤린 공급 업체
글로벌( 12)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2503 | 58 |
| PUSHAN INDUSTRIAL (SHAANXI) CO.,LTD | +8618291886785 |
olivia.wang@pushanshiye.com | China | 164 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-81138252 +86-18789408387 |
1057@dideu.com | China | 3406 | 58 |
| Neostar United (Changzhou) Industrial Co., Ltd. | +86-519-519-85557386 |
marketing1@neostarunited.com | China | 11506 | 58 |
| ShenZhen Trendseen Biological Technology Co.,Ltd. | 13417589054 |
trendseenbio@gmail.com | China | 11681 | 58 |
| SUZHOU SENFEIDA CHEMICAL CO.,LTD | +86-0512-83500002 +8615195660023 |
sales@senfeida.com | China | 23053 | 58 |
| Shanghai Eyougene biotechnology development co.LTD | 021-52277372 13564545227 |
dom.sales@eyougene.com | China | 245 | 58 |
| Shanghai Famo Bio-chemical Technology Company Ltd. | 021-36680027 15821745250 |
fengqiang@famobiotech.com | China | 528 | 56 |
| Shaanxi Dideu Medichem Co. Ltd | 029-81118914 18066712840 |
1027@dideu.com | China | 10011 | 58 |
| Pushan Industry (Shaanxi) Co., Ltd. | 029-81310890 13571859809 |
info@pushanshiye.com | China | 9970 | 58 |