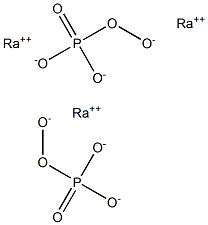
Radium Peroxyphosphate
|
|
Radium Peroxyphosphate 속성
안전
Radium Peroxyphosphate C화학적 특성, 용도, 생산
개요
Radium peroxyphosphate has remained unknown and no publications concerning this salt are available in the scientific literature. This is not surprising in view of the intense radioactivity of 226Ra. Like the other alkaline earths, radium peroxyphosphates could be prepared by both solid-state or solution methods. For the monoperoxyphosphates, one would get RaHPO5 for the former method. The question of degree of hydration remains unknown.In the solution method, one could use the acid, H3PO5 or the potassium salt K2HPO5. Whether the solution should be acidic or basic is not known. Like the other alkaline earths, it is probably best to prepare the acid in a nonaqueous solution like carbon tetrachloride and then add the potassium compound for the basic solution:
P2O5 + 2H2O2 +H2O ? 2H3PO5
2H3PO5 + KOH ? K2HPO5
To either of these is added the radium compound to form the monoperoxyphosphate:
Ra(OH)2 + K2PO50RaHPO5 +H2O
To prepare radium diperoxy compounds, the potassium monoperoxyphosphate compound is transformed to the diperoxyphosphate compound, in a nonaqueous solution at low temperature:
K2HPO5 + P2O5 ? K2P2O8
Radium Peroxyphosphate 준비 용품 및 원자재
원자재
준비 용품
Radium Peroxyphosphate 공급 업체
글로벌( 0)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|
Radium Peroxyphosphate 관련 검색:
Beryllium Peroxyphosphate
Radium Metaphosphates
Strontium Hypophosphate
Magnesium Peroxyphosphate
Calcium Hypophosphate
Magnesium hypophosphate
Barium Peroxyphosphate
Strontium Peroxyphosphate
BARIUM METAPHOSPHATE
Calcium Perphosphate
Radium Hypophosphate
Beryllium Hypophosphate
Barium Hypophosphate