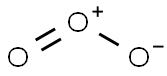
오존
|
|
오존 속성
- 녹는점
- 193℃
- 끓는 점
- -110℃
- 밀도
- 1.46 g/cm3
- 증기압
- 55kPa at -12℃
- 용해도
- slightly soluble in H2O
- 물리적 상태
- 청색 가스
- 색상
- 파란색 또는 보라색-검정색 고체 또는 불안정한 무색 가스 또는 진한 파란색 액체
- 냄새
- 자극적인 냄새, 0.01~0.04ppm에서 감지 가능; 1ppm에서는 날카롭고 불쾌한 냄새가 납니다.
- Odor Threshold
- 0.0032ppm
- 수용성
- 570mg/L at 20℃
- 노출 한도
- TLV-TWA 0.1 ppm (~0.2 mg/m3) (ACGIH, NIOSH, and MSHA), 0.2 ppm (~0.4 mg/m3) (OSHA); IDHL 10 ppm (NIOSH).
- 안정성
- Unstable - may decompose spontaneously and violently to oxygen. Mixtures containing a moderate partial pressure of ozone, and pure ozone at even low pressures are both potentially explosive. May react very violently with combustible materials and reducing agents, such as organics. Even small quantities of organic material, such as traces of g
- LogP
- -0.87 at 20℃
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 유엔번호(UN No.) | 1956 | ||
|---|---|---|---|
| OEL | Ceiling: 0.1 ppm (0.2 mg/m3) | ||
| 위험 등급 | 2.2 | ||
| 유해 물질 데이터 | 10028-15-6(Hazardous Substances Data) | ||
| 독성 | LC50 inhal (rat) 4.8 ppm (4 h) PEL (OSHA) 0.1 ppm (0.2 mg/m3) TLV-TWA (ACGIH) 0.1 ppm (0.2 mg/m3) STEL (ACGIH) 0.3 ppm (0.6 mg/m3) |
||
| IDLA | 5 ppm | ||
| 기존화학 물질 | KE-27742 |
| 그림문자(GHS): |
    
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 신호 어: | Danger | ||||||||||||||||||||||||||||||||||||||||||||||||||
| 유해·위험 문구: |
|
||||||||||||||||||||||||||||||||||||||||||||||||||
| 예방조치문구: |
|
오존 C화학적 특성, 용도, 생산
화학적 성질
Ozone,O3, a colorless gas or dark blue liquid, also known as activated oxygen, is an allotropic form of oxygen formed in nature by lightning in air and during evaporation of water particularly by spray in the sea.It is an unstable blue gas with a distinctive odor. Condenses to a blue black liquid or crystalline solid. Ozone absorbs ultraviolet rays and acts as a natural blanket that protects the earth from harmful short-wave radiation from the sun. Ozone is a powerful oxidizer. It is used as an oxidant in the rubber industry, as a bleaching agent,as a water purifier, and to treat industrial wastes.
물리적 성질
Ozone is an allotropic molecular form of oxygen containing three atoms of oxygen (O3).It is a much more powerful oxidizing agent than diatomic oxygen (O2) or monatomic oxygen(O). It is the second most powerful oxidizer of all the elements. Only fluorine is a strongeroxidizer. It is not colorless as is oxygen gas. Rather, ozone is bluish in the gaseous state, butblackish-blue in the liquid and solid states (similar to the color of ink).Ozone’s boiling point is –112°C, and its freezing point is –192°C.
Origin of Name
From the Greek words oxys (which means sharp or acid) and gen (which means forming); together they stand for “acid-forming.” In the eighteenth century, it was believed that all acids contained oxygen.역사
It was once believed that air was a single element, but by the fifteenth century ce, scientistsbegan to question whether it was possibly at least two separate gases. Leonardo da Vinci wasone of the first to suggest the air consisted of at least two gases. He even determined that oneof them would support life and fire.In 1839 Christian Friedrich Schonbein (1799–1868) discovered a gas with an unusualodor coming from some electrical equipment. He did not know what it was, but because ithad an odd smell, he called it “ozone,” after the Greek word for “I smell.” Although he knewthat it was a chemical substance, he mistakenly associated ozone with the halogens (group 17).Others before Schonbein had smelled the gas but had not recognized its importance. ThomasAndrews (1813–1885) and several other scientists, through different experiments, identifiedozone as a form of oxygen (an allotrope). It was not until 1868 that J. Louis Soret establishedthe formula to be O3.
용도
Ozone is used as an oxidizing compound, as a disinfectant for air and water, for bleaching waxes and oil, and in organic synthesis. It occurs in the atmosphere at sea level to about 0.05 ppm. It is produced by the action of ultraviolet (UV) radiation on oxygen in air.정의
A poisonous, blue-colored allotrope of oxygen made by passing oxygen through a silent electric discharge. Ozone is unstable and decomposes to oxygen on warming. It is present in the upper layers of the atmosphere, where it screens the Earth from harmful short-wave ultraviolet radiation. There is concern that the ozone layer is possibly being depleted by the use of fluorocarbons and other compounds produced by industry.제조 방법
Ozone is generated from oxygen by passing an electric spark or silent electrical discharge through dry, and pure oxygen. This electrical discharge may be applied between two glass surfaces between which oxygen is passed. Many types of ozonizers (ozone generating apparatus) are known and commercially available for small-scale production of this gas for various uses. Ozone may be produced by electrolysis of chilled dilute sulfuric acid (e.g. 2.5N H2SO4) or perchloric acid at high current density (higher than that required to produce oxygen alone). A mixture of oxygen and ozone evolve at the anode.생산 방법
Ozone (triatomic oxygen) is a light blue gas with a characteristic odor (reminiscent to some individuals of an electrical discharge such as lightening). Ozone was first described in 1840 by Christian Friedrich Schonbein [1799–1868], who produced it from phosphorus and electrolysis of water. Schonbein also developed a colorimetric assay involving starch and potassiumiodide-impregnated paper that was widely used to measure atmospheric ozone concentrations. Interestingly, Schonbein’s studies were interrupted when he discovered the acute toxicity of ozone in 1851 and noted that ozone caused “a really painful affection of the chest, a sort of asthma, connected with a violent cough”. Concern of ozone’s toxicity dates back to the mid-twentieth century, when it was recognized as a major air pollutant in urban areas. Additional concerns arose in the 1980s and 1990s regarding its depletion in the stratosphere.Ozone can be found naturally in the troposphere during electrical storms and in the stratosphere. Background levels of ozone in nonurban areas average about 10–20 ppb and are due mainly to intrusion of stratospheric ozone into the lower atmosphere.
화학 반응
Ozone reacts (1) with potassium iodide, to liberate iodine, (2) with colored organic materials, e.g., litmus, indigo, to destroy the color, (3) with mercury, to form a thin skin of mercurous oxide causing the mercury to cling to the containing vessel, (4) with silver film, to form silver peroxide, Ag2O2, black, produced most readily at about 250 C, (5) with tetramethyldiaminodiphenylmethane (CH3)2N·C6H4·CH2·C6H4·N(CH3)2, in alcohol solution with a trace of acetic acid to form violet color (hydrogen peroxide, colorless; chlorine or bromine, blue; nitrogen tetroxide, yellow). In contrast to hydrogen peroxide, ozone does not react with dichromate, permanganate, or titanic salt solutions. Ozone reacts with olefin compounds to form ozonide addition compounds. Ozonides are readily split at the olefinin-ozone position upon warming alone, or upon warming their solutions in glacial acetic acid, with the formation of aldehyde and acid compounds which can be readily identified, thus serving to locate the olefin position in oleic acid, C17H33·COOH, as midway in the chain (CH3(CH2)7CH:CH(CH2)7COOH. Ozone is used (1) as a bleaching agent, e.g., for fatty oils, (2) as a disinfectant for air and H2O, (3) as an oxidizing agent.일반 설명
A colorless to bluish gas that condenses to a dark blue liquid, or blue-black crystals. Has a characteristic odor in concentrations less than 2 ppm. Used as a disinfectant for air and water; used for bleaching waxes, textiles and oils, ozonolysis of unsaturated fatty acids to pelargonic and other acids; manufacture of ink; catalyst; water treatment for taste and odor control; mold and bacteria inhibitor in cold storage; bleaching agent.반응 프로필
Ozone is a propellant; ignites upon contact with alcohols, amines, ammonia, beryllium alkyls, boranes, dicyanogen, hydrazines, hydrocarbons, hydrogen, nitroalkanes, powdered metals, silanes, or thiols [Bretherick 1979. p.174]. Aniline in a atmosphere of Ozone produces a white galatinous explosive ozobenzene [Mellor 1:911. 1946-47]. A mixture of ether and Ozone forms aldehyde and acetic acid and a heavy liquid, ethyl peroxide, an explosive [Mellor 1:911. 1946-47]. Severe explosions occur attempting to form tribromic octaoxide from bromine and Ozone [Mellor 2, Supp. 1:748. 1956]. Mixtures of Ozone and dinitrogen pentaoxide are flammable or explosive [Mellor 8, Supp. 2:276. 1967]. Ozone and ethylene react explosively [Berichte 38:3837]. Nitrogen dioxide and Ozone react with the evolution of light, and often explode [J. Chem. Phys. 18:366 1920]. Contact of very cold liquefied gas with water may result in vigorous or violent boiling of the product and extremely rapid vaporization due to the large temperature differences involved. If the water is hot, there is the possibility that a liquid "superheat" explosion may occur. Pressures may build to dangerous levels if liquid gas contacts water in a closed container, [Handling Chemicals Safely 1980].위험도
High concentrations of ozone are a fire and explosion hazard when in contact with anyorganic substance that can be oxidized.In moderately high concentrations ozone is very toxic when inhaled, and in lesser concentrations,it is irritating to the nose and eyes. Ozone in the lower atmosphere contributes to airpollution and smog. It can cause damage to rubber, plastics, and paints. These low concentrationscan cause headaches, burning eyes, and respiratory irritation. It is particular harmful toasthmatics and the elderly with respiratory problems.
건강위험
Ozone is a highly toxic gas that is extremely irritating to the eyes, mucous membranes, and respiratory tract. The characteristic odor of ozone can be detected below the permissible exposure limit, and this compound is therefore regarded to have adequate warning properties. However, at higher concentrations the ability to smell ozone may decrease. Inhalation of 1 ppm ozone may cause headaches and irritation of the upper and lower respiratory tract. The first symptoms of exposure include irritation of the eyes, dryness of throat, and coughing; these symptoms disappear after exposure ceases. Exposure at higher levels may lead to lacrimation, vomiting, upset stomach, labored breathing, lowering of pulse rate and blood pressure, lung congestion, tightness in the chest, and pulmonary edema, which can be fatal. Exposure to 100 ppm of ozone for 1 hour can be lethal to humans. Animal studies indicate that chronic exposure to ozone may result in pulmonary damage, leading to chronic lung impairment. Continual daily exposure to ozone can cause premature aging.화재위험
Severe explosion hazard when shocked, exposed to heat or flame, or by chemical reaction with organic substances, especially reducing agents. Ozone is a powerful oxidizing agent. Incompatible with alkenes; aromatic compounds; benzene, rubber; bromine; dicyanogen; diethyl ether; dinitrogen tetroxide; hydrogen bromide; 4-hydroxy-4-methyl-1,6-heptadiene; nitrogen trichloride; stibine; tetrafluorohydrazine. Avoid contact with organic materials.인화성 및 폭발성
Ozone by itself is not flammable. Liquid ozone and concentrated solutions are extremely hazardous and can explode on warming or when shocked.잠재적 노출
Ozone is found naturally in the atmosphere as a result of the action of solar radiation and electrical storms. It is also formed around electrical sources, such as X-ray or ultraviolet generators, electric arcs; mercury vapor lamps; linear accelerators; and electrical discharges. Ozone is used as an oxidizing agent in the organic chemical industry (e.g., production of azelaic acid); as a disinfectant for air, mold and bacteria inhibitor for food in cold storage rooms, and for water (e.g., public water supplies; swimming pools; and sewage treatment); for bleaching textiles; waxes, flour, mineral oils, and their derivatives; paper pulp; starch, and sugar; for aging liquor and wood; for processing certain perfumes; vanillin, and camphor; in treating industrial wastes; in the rapid drying of varnishes and printing inks; and in the deodorizing of feathers.Carcinogenicity
Ozone has been positive as a genotoxic substance in certain assay systems, but the results are inconsistent. For example, in vitro assays have noted that ozone can induce bacterial mutations, plasmid DNA strand breakage, chromatid and chromosome aberrations in lymphocytes, and a doubling of the frequency of preneoplastic variants compared with control cultures. However, in vivo assays of similar end points produced mixed results. For example, alveolar macrophages from rats exposed to 270–800 ppb ozone developed chromatid damage, but no chromosomal changes. In human subjects exposed to 500 ppb ozone (6–10 h), a slight increase in sister chromatid exchange persisted for ≤6 weeks. In contrast, no significant changes in chromosome or chromatid breaks were observed in lymphocytes of subjects exposed for 4 h to 400 ppb. Cultured human epidermal cells exposed to 500 ppb ozone for 10 min showed no evidence of DNA strand breakage.Other investigators have suggested that chronic ozone exposure may facilitate the development of benign pulmonary tumors (adenomas) in mice and other hyperplastic nodules in the lungs of nonhuman primates. As is true of hyperoxia, ozone exposure may enhance or retard lung tumorigenesis by other agents in rodents, depending on the exposure protocol.
Other investigators have suggested that in vitro assays indicate ozone may exert indirect genotoxic effects. Ozone has been purported to affect the integrity of immune system defenses against tumor development and progression (1073, 1074). In addition, arylamines found in tobacco smoke (e.g., naphthylamine and toluidine isomers) can be chemically altered by brief exposures (1 h) to 100–400 ppb ozone. The unidentified stable products of this reaction cause single-strand DNA breaks in cultured human lung cells equivalent to that produced by 100 rad of irradiation. However, an in vivo cocarcinogenicity study failed to find similar effects.
환경귀착
Ozone formed from anthropogenic sources such as from car vehicle emissions in the troposphere can travel long distances. Ozone formation and scavenging by other chemicals such as NO is in constant daily flux. There are times when solar radiation is high, such as on hot days or during rush hour, which produces elevated ozone levels and times such as during the evening when the rate of ozone scavenging exceeds ozone production, resulting in less ozone in the atmosphere. Ozone concentrations in the eastern United States are often more than 80 ppb in the warm spring and summer months, though ozone levels in the western United States are lower.저장
Work with ozone should be conducted in a fume hood to prevent exposure by inhalation. Ozone is usually produced in the laboratory with a ozone generator, and care should be taken to ensure adequate ventilation in the area where the ozone generation equipment is located. Because of the possibility of the generation of explosive ozonides, ozonolysis reactions should always be conducted in a fume hood behind a safety shield.운송 방법
UN1955 Compressed gas, toxic, n.o.s, Inhalation Hazard Zone A, Hazard Class: 2.3; Labels: 2.3-Poisonousgas, 5.1-Oxidizer, Technical name required, Inhalation Hazard Zone A. Cylinders must be transported in a secure upright position, in a well-ventilated truck. Protect cylinder and labels from physical damage. The owner of the compressed gas cylinder is the only entity allowed by federal law (49CFR) to transport and refill them. It is a violation of transportation regulations to refill compressed gas cylinders without the express written permission of the owner.비 호환성
A powerful oxidizer. A severe explosion hazard when exposed to shock or heat, especially solid or liquid form. Spontaneously decomposes to oxygen under ordinary conditions; heating increases oxygen production. Reacts with all reducing agents; combustibles, organic, and inorganic oxidizable materials; and can form products that are highly explosive. Incompatible with alkenes, aniline, benzene, bromine, ether, ethylene, and hydrogen bromide; nitric oxide; stibine. Attacks metals except gold and platinum.폐기물 처리
Vent to atmosphere. Use a licensed professional waste disposal service to dispose of this material. All federal, state, and local environmental regulations must be observed. Return refillable compressed gas cylinders to supplier.오존 준비 용품 및 원자재
원자재
준비 용품
오존 공급 업체
글로벌( 19)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21622 | 55 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-89586680 +86-18192503167 |
1026@dideu.com | China | 7922 | 58 |
| XIAMEN AMITY INDUSTRY AND TRADE CO., LTD. | +8618950047208 |
ellena@amitychem.com | China | 43416 | 58 |
| Mainchem Co., Ltd. | +86-0592-6210733 |
sale@mainchem.com | China | 32343 | 55 |
| Explore the platform | 400-111-6333 |
180830201name@qq.com | CHINA | 6730 | 58 |
| Beijing Putian Tongchuang Biotechnology Co., Ltd. | 18153089275 |
3032079623@qq.com | China | 6695 | 58 |
오존 관련 검색:
로다민 B 포스포몰리브드 산 수화물 오존
OZONIZED OLIVE OIL
Ninhydrine, ozone friendly ready to use spray for TLC chromatography
Sulfur dioxide mixed with ozone (1:1)
DIMETHYLAMINOBENZALDEHYDE, OZONE FRIENDLY READY TO USE SPRAY FOR TLC
BROMOCRESOL GREEN, OZONE FRIEND.READY TO USE SPRAY FOR TLC IN 2-PROH, PURE,Bromocresol Green, ozone friendly ready to use TLC spray in 2-PrOH, pure
OZONIZED SUNFLOWER SEED OIL
SET OF ALR-CFCS
OZONIZED JOJOBA OIL
NINHYDRIN, OZONE FRIENDLY READY TO USE SPRAY FOR TLC
OZONIZED CASTOR OIL
DUST-PRO PRESSURIZED DUSTER OZONE-
DUST-OFF PLUS VALVE PACKAGED IN PLASTIC CARD, OZONE FREE SOME FLAMMABLES
OZONE OXYGEN
OZONE-DEPLETINGCHEMICALS
OZONE REAGENT