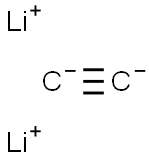디리튬아세틸리드 C화학적 특성, 용도, 생산
화학적 성질
crystal(s) white powder(s); decomposes in water, evolves acetylene when dissolved in acid [HAW93]
제조 방법
Lithium carbide, Li2C2, may be prepared by the direct combination of lithium and
carbon at about 1000°C or by the reaction of lithium metal with acetylene in liquid ammonia.
The latter process first yields lithium acetylide, LiC=CH, which decomposes to lithium
carbide. The conversion to the carbide is incomplete, however . A convenient laboratory
preparation of lithium carbide may be carried out by bubbling acetylene into a solution of
n-butyllithium in hexane. The precipitated carbide may be filtered from the reaction
mixture, washed, and dried. Lithium carbide is ionic and yields acetylene on hydrolysis.
디리튬아세틸리드 준비 용품 및 원자재
원자재
준비 용품