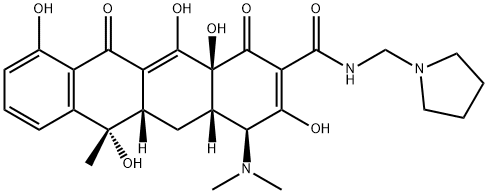
ROLITETRACYCLINE
|
|
ROLITETRACYCLINE 속성
- 녹는점
- 163.5°C
- 끓는 점
- 608.16°C (rough estimate)
- 밀도
- 1.2902 (rough estimate)
- 굴절률
- 1.7120 (estimate)
- 저장 조건
- -20°C Freezer, Under inert atmosphere
- 용해도
- DMSO(약간 용해됨), 메탄올(매우 소량)
- 물리적 상태
- 고체
- 물리적 상태
- 단단한 모양
- 산도 계수 (pKa)
- pKa 7.4 (Uncertain)
- 색상
- 매우 어두운 빨간색에서 매우 어두운 갈색까지
- 수용성
- >20g/L(21ºC)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 22-36/37/38 | ||
| 안전지침서 | 26-36 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | QI9150000 |
ROLITETRACYCLINE C화학적 특성, 용도, 생산
개요
Rolitetracycline was synthesized in 1958. Bristol-Myers Laboratories prepared it from tetracycline by introducing a pyrrolidinylmethyl moiety. This antibiotic is very soluble in water and more stable than tetracycline under acidic conditions. Rolitetracycline is used by intravenous injection, and its nitrate is used by both intravenous and intramuscular injections for therapy of the same infections as those treated by tetracycline.용도
Rolitetracycline is a tetracycline antibiotic that may be given intravenously or intramuscularly in serious bacterial infections when oral administration is not practicable.정의
ChEBI: A derivative of tetracycline in which the amide function is substituted with a pyrrolidinomethyl group.일반 설명
Rolitetracycline, N-(pyrrolidinomethyl)tetracycline(Syntetrin), was introduced for use by intramuscular or intravenousinjection. This derivative is made by condensingtetracycline with pyrrolidine and formaldehyde in the presenceof tert-butyl alcohol. It is very soluble in water (1 g dissolvesin about 1 mL) and provides a means of injecting theantibiotic in a small volume of solution. It has been recommendedfor cases when the oral dosage forms are not suitable,but it is no longer widely used.Pharmaceutical Applications
2-N-pyrrolidinomethyl-tetracycline. A semisynthetic derivative of tetracycline supplied as the nitrate sesquihydrate for parenteral use.It is not absorbed from the gastrointestinal tract. It is highly soluble and therefore can be administered parenterally. Peak plasma concentrations of 4–6 mg/L occur at 0.5–1 h after 350 mg intravenously. The plasma elimination half-life is 5–8 h. About 50% of the dose is excreted in the urine, producing high concentrations.
Intravenous administration is occasionally accompanied by abnormal taste, shivering and rigors, hot flushes, facial reddening, dizziness and, rarely, circulatory collapse. Symptoms of myasthenia gravis have occasionally been exacerbated.
ROLITETRACYCLINE 준비 용품 및 원자재
원자재
준비 용품
ROLITETRACYCLINE 공급 업체
글로벌( 45)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| InvivoChem | +1-708-310-1919 +1-13798911105 |
sales@invivochem.cn | United States | 6393 | 58 |
| Shanghai Acmec Biochemical Technology Co., Ltd. | +undefined18621343501 |
product@acmec-e.com | China | 33350 | 58 |
| Aladdin Scientific | +1-+1(833)-552-7181 |
sales@aladdinsci.com | United States | 52927 | 58 |
| J & K SCIENTIFIC LTD. | 010-82848833 400-666-7788 |
jkinfo@jkchemical.com | China | 96815 | 76 |
| Chemsky(shanghai)International Co.,Ltd. | 021-50135380 |
shchemsky@sina.com | China | 32344 | 50 |
| Spectrum Chemical Manufacturing Corp. | 021-021-021-67601398-809-809-809 15221380277 |
marketing_china@spectrumchemical.com | China | 9664 | 60 |
| BOC Sciences | 1-631-485-4226; 16314854226 |
info@bocsci.com | United States | 14059 | 65 |
| Shanghai TaoSu Biochemical Technology Co., Ltd. | 021-33632979 |
info@tsbiochem.com | China | 8073 | 58 |
| Beijing Jin Ming Biotechnology Co., Ltd. | 010-60605840 18892239720 |
psaitong@jm-bio.com | China | 12308 | 58 |
| Lynnchem | 86-(0)29-85992781 17792393971 |
info@lynnchem.com | China | 4587 | 58 |