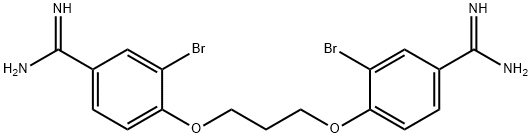
디브롬프로파미딘
|
|
디브롬프로파미딘 속성
안전
디브롬프로파미딘 C화학적 특성, 용도, 생산
Originator
Dibrompropamidine,Aventis Pharma ASManufacturing Process
2,2’-Dibromo-4,4’-dicyano-α,γ-diphenoxypropane (3.0 g) and anhydrous ethanol (3.0 ml) were dissolved in anhydrous chloroform (40 ml). The solution was saturated at 0°C with dry hydrogen chloride and set aside for 7 day. The iminoether hydrochloride which crystallised out, was filtered off and washed with light petroleum. The solid was added to 12% ammoniacal ethanol (47 c.c.) and the mixture was heated at 60°C for 6 hours. Solution was obtained after 3 hours and the amidine hydrochloride began to cryistallise. The mixture was ice-cooled, and the white crystalline solid was filtered off, washed with 2 N hydrochloric acid, and recrystallised twice from 0.5 N hydrochloric acid.2,2'-Dibromo-4,4'-diamidino-α,γ-diphenoxypropane dihydrochloride (2.1 g) [another name: 4,4'-(Trimethylenedioxy)bis(3-bromobenzamidine)] separated as white prisme. Melting point 309°C. (decomp.).
Berg S.S., GB Patent No. 598,911; March 22, 1945; Assigned to May and Bakker Limited, a British Co., Dagenham, Essecs.
Therapeutic Function
Antiprotozoal디브롬프로파미딘 준비 용품 및 원자재
원자재
준비 용품
디브롬프로파미딘 공급 업체
글로벌( 7)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hangzhou MolCore BioPharmatech Co.,Ltd. | +86-057181025280; +8617767106207 |
sales@molcore.com | China | 49739 | 58 |
| Hangzhou Synstar pharmaceutical Technology CO.,Ltd | 0571-85361029 |
synstar518@163.com | China | 1991 | 58 |
| Hubei Qingbei Yunyan Pharmaceutical Technology Co., Ltd | 18162595016 |
3287908757@qq.com | China | 9131 | 58 |