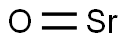스트론튬 산화물
|
|
스트론튬 산화물 속성
- 녹는점
- 2430°C
- 끓는 점
- 3000°C
- 밀도
- 4.7 g/mL at 25 °C (lit.)
- 인화점
- 3000°C
- 저장 조건
- Sealed in dry,Room Temperature
- 용해도
- 수산화칼륨 용액에 용해됩니다. 알코올에 약간 용해됩니다. 아세톤과 에테르에 용해되지 않습니다.
- 물리적 상태
- 가루
- Specific Gravity
- 4.7
- 색상
- 하얀색
- 수용성
- 융합된 수산화칼륨에 용해됩니다. 에테르와 아세톤에 불용성. 열이 발생하면서 물에서 Sr(OH){2}로 분해됩니다. 알코올에 약간 용해됩니다. 아세톤과 에테르에 용해되지 않습니다.
- Crystal Structure
- NaCl type
- 감도
- Moisture Sensitive
- Merck
- 14,8846
- crystal system
- Cube
- Space group
- Fm3m
- Lattice constant
a/nm b/nm c/nm α/o β/o γ/o V/nm3 0.5113 0.5113 0.5113 90 90 90 0.1337
- InChI
- InChI=1S/O.Sr
- InChIKey
- UFQXGXDIJMBKTC-UHFFFAOYSA-N
- SMILES
- O=[Sr]
- CAS 데이터베이스
- 1314-11-0(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | C | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 14-34 | ||
| 안전지침서 | 26-30-36/37 | ||
| 유엔번호(UN No.) | UN 3262 8/PG 2 | ||
| WGK 독일 | 3 | ||
| TSCA | Yes | ||
| 위험 등급 | 8 | ||
| 포장분류 | II | ||
| 기존화학 물질 | KE-32238 |
스트론튬 산화물 C화학적 특성, 용도, 생산
개요
Strontium Oxide, SrO, is a metal oxide usually produced by the reaction of strontium with oxygen. It can also be prepared by the decomposition of heated strontium carbonate, hydroxide, or nitrate and is used in manufacturing other strontium salts, in pigments, soaps and greases, and as a drying agent. It is commonly used in the glass, ceramics, and electronic industries and has other specific uses, such as fuel cells and sputtering target material. If burn strontium under general conditions with air, strontium oxide and strontium nitride will form. Strontium oxide was once required by law as the material used on old TV face-plates to block harmful X-ray emissions. BaO and SrO were reported for the transesterification of palm oil for the production of biodiesel. The reaction was conducted under ultrasound conditions to ensure the effective production of biodiesel.화학적 성질
white to grayish white; reacts with water, forming Sr(OH)2, with evolution of heat; enthalpy of fusion 75.00kJ/mol [MER06] [CRC10]물리적 성질
Strontium oxide or strontia, SrO, is formed when Sr metal reacts with oxygen:2Sr+ O2→2SrO
Burning strontium in air results in a mixture of strontium oxide and strontium nitride. It also forms from the decomposition of strontium carbonate SrCO3. It is a strongly basic oxide. It reacts with moisture forming the hydroxide and with carbon dioxide in the air to form the carbonate.
용도
SrO is also used in medicine, pyrotechnics, pigments, greases, soaps, and as a chemical intermediate. It is also produced as high-purity strontium oxide rotatable sputtering targets with the highest possible density and smallest possible average grain sizes for use in semiconductor, photovoltaic, and coating applications by chemical vapor deposition and physical vapor deposition and optical applications.One of the reasons why rare metals are restrained from wide use in production is their high cost.스트론튬 산화물 준비 용품 및 원자재
원자재
준비 용품
스트론튬 산화물 공급 업체
글로벌( 197)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Alfa Chemistry | |
Info@alfa-chemistry.com | United States | 24072 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-81148696 +86-15536356810 |
1022@dideu.com | China | 3878 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21663 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29888 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 |
sales@chemdad.com | China | 39916 | 58 |
| Antai Fine Chemical Technology Co.,Limited | 18503026267 |
info@antaichem.com | CHINA | 9641 | 58 |
| SIMAGCHEM CORP | +86-13806087780 |
sale@simagchem.com | China | 17367 | 58 |
| Hubei Ipure Biology Co., Ltd | +8613367258412 |
ada@ipurechemical.com | China | 10326 | 58 |
스트론튬 산화물 관련 검색:
스트론튬 수산화물 스트론튬 산화물 스트론슘 FERRITE 비스머스 스트론튬 칼슘 구리 옥사이드 산화 스트론슘 티타늄 스트론튬 과산화스트론듐 납이 섞인 산화바륨, 고체 용액과 산화칼슘, 산화스트론튬, 산화텅스텐 Boric acid, solid soln. with barium oxide, calcium oxide and strontium oxide, lead and manganese-doped
TITANIUM DIOXIDE
Tetraterbium heptaoxide
Praseodymium oxide
Rhodium oxide
MANGANESE (III) OXIDE
Europium Oxide
COPPER CHROMITE
Cobalt oxide
Gadolinium oxide