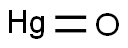Mercuric Oxide suppliers
Mercuric Oxide
- CAS:
- 21908-53-2
- MF:
- HgO
- MW:
- 216.59
Properties
- Melting point:
- 500 °C (dec.)(lit.)
- Density
- 11,14 g/cm3
- storage temp.
- Poison room
- solubility
- Aqueous Acid (Slightly), Methanol (Very Slightly)
- form
- Powder
- Specific Gravity
- 11.14
- color
- Yellow to orange
- Odor
- Odorless
- Water Solubility
- Partially soluble in water. insoluble in alcohol, ether, acetone and ammonia.
- Merck
- 14,5882
- Stability:
- Stable. Sensitive to light. Incompatible with strong oxidizing agents, strong reducing agents, combustible materials, organic materials, phenols.
Safety Information
- Symbol(GHS)



GHS06,GHS08,GHS09
- Signal word
- Danger
- Hazard statements
- H300+H310+H330-H373-H410
- Precautionary statements
- P262-P273-P280-P301+P310+P330-P302+P352+P310-P304+P340+P310
- Hazard Codes
- T+,N
- Risk Statements
- 26/27/28-33-50/53
- Safety Statements
- 13-28-45-60-61-28A
- RIDADR
- UN 1641 6.1/PG 2
- WGK Germany
- 3
- RTECS
- OW8750000
- F
- 8
- TSCA
- Yes
- HazardClass
- 6.1
- PackingGroup
- II
Use
MERCURIC OXIDE, RED is highly toxic by ingestion, inhalation, or skin absorption. Very short exposure to small quantities may cause death or permanent injury. Following ingestion, mercuric oxide is readily converted to mercuric chloride, the most dangerous mercury compound. Mercuric oxide dust has a corrosive effect on eyes, skin, and respiratory tract. People with a history of allergies or known sensitization to mercury, chronic respiratory disease, nervous system disorders, or kidney disorders are at increased risk from exposure.
According to relevant laws, regulations, policies and the provisions of this website, this website does not display the product information!
The source is "剧毒、有毒、易致癌化学品". If the information is inaccurate, please contact the platform to modify it.
contact information:info@chemicalbook.com 400-158-660