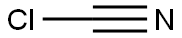Cyanogen chloride suppliers
Cyanogen chloride
- CAS:
- 506-77-4
- MF:
- CClN
- MW:
- 61.47
Properties
- Melting point:
- -6°
- Boiling point:
- 14°C
- Density
- 1.186
- vapor pressure
- 60kPa at 0℃
- solubility
- soluble in H2O, ethanol, ethyl ether
- form
- colorless gas
- color
- colorless vol liquid or gas
- Water Solubility
- soluble H2O, ether, alcohol [MER06]
- Exposure limits
- Ceiling limit 0.7 mg/m3 (0.3 ppm) (ACGIH).
- LogP
- 0.64 at 20℃
Safety Information
- Symbol(GHS)




GHS09,GHS05,GHS04,GHS06
- Signal word
- Danger
- Hazard statements
- H319-H280-H300+H310+H330-H400-H314
- Precautionary statements
- P410+P403-P264-P280-P305+P351+P338-P337+P313P-P273-P391-P501-P260-P264-P280-P301+P330+P331-P303+P361+P353-P363-P304+P340-P310-P321-P305+P351+P338-P405-P501
- Hazard Codes
- T
- RIDADR
- UN 1589
- OEL
- Ceiling: 0.3 ppm (0.6 mg/m3)
- HazardClass
- 2.3
- Toxicity
- CK may polymerize violently if contaminated with hydrogen chloride or ammonium chloride. Upon heating, it decomposes, producing toxic and corrosive fumes of hydrogen cyanide, hydrochloric acid, and nitrogen oxides. Hazardous polymerization can occur. Cyanogen chloride will react slowly with water to form hydrogen chloride gas. It acts on the body in two ways. Systemic effects of CK are much like those of hydrogen cyanide. Additionally, it causes irritation of the eyes, upper respiratory tract, and lungs. Eye irritation results in tearing. CK, like AC, stimulates the respiratory system and rapidly paralyzes it. Exposure is followed by immediate, intense irritation of the nose, throat, and eyes, with coughing, tightness in the chest, and lacrimation. This is followed by dizziness and increasing difficulty breathing. Unconsciousness comes next, with failing respiration and death within a few minutes. Convulsions, retching, and involuntary urination and defecation may occur. If these effects are not fatal, the signs and symptoms of pulmonary edema may develop. There may be repeated coughing, with profuse foamy sputum, rales in the chest, severe dyspnea, and distinct cyanosis. Recovery from the systemic effects is usually as prompt as in AC poisoning. However, a higher incidence of residual damage to the central nervous system should be expected. Based upon the concentration of the cyanogen chloride to which the victim has been exposed, the pulmonary effects may evolve instantly or may be delayed until the systemic effects have subsided. Consequently, early prognosis must be cautious.
Use
Cyanogen chloride is a highly poisonous compound and a severe irritant. In humans, exposure to 1 ppm for 10 minutes caused severe irritation of eyes and nose. Irritation of respiratory tract is followed by hemorrhage of the bronchi and trachea, as well as pulmonary edema. The toxicity of cyanogen chloride is attributed to its relatively easy decomposition to cyanide ion in an aqueous medium. The cyanide attacks the cells in the body and interferes with the cellular metabolism. Tests on rats indicated that exposure to cyanogen chloride caused lacrimation and chronic pulmonary edema and somnolence. At a high concentration of 300 ppm, death occurred to the test animals. Inhalation of 48 ppm for 30 minutes was fatal to humans. In animals, subcutaneous intakes of 5 and 15 mg/kg were lethal to both dogs and rabbits. Chronic exposure to cyanogen chloride can cause conjunctivitis and edema of the eyelid.
According to relevant laws, regulations, policies and the provisions of this website, this website does not display the product information!
The source is "剧毒、有毒、易致癌化学品". If the information is inaccurate, please contact the platform to modify it.
contact information:info@chemicalbook.com 400-158-660