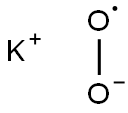Potassium superoxide suppliers
Potassium superoxide
- CAS:
- 12030-88-5
- MF:
- KO2*
- MW:
- 71.1
Properties
- Melting point:
- 400 °C
- Density
- 2,14 g/cm3
- storage temp.
- Store at +15°C to +25°C.
- solubility
- Soluble in ethanol and ether.
- form
- powder
- color
- Yellow-green
- Specific Gravity
- 2.14
- PH
- >7 (H2O, 20℃)
- Water Solubility
- reacts
- Sensitive
- Air & Moisture Sensitive
- Stability:
- Stable, but reacts violently with water. Incompatible with moisture, alcohols, strong reducing agents, strong acids, finely powdered metals.
Safety Information
- Symbol(GHS)


GHS03,GHS05
- Signal word
- Danger
- Hazard statements
- H271-H314
- Precautionary statements
- P220-P280-P305+P351+P338-P310
- Hazard Codes
- O,C
- Risk Statements
- 8-14-34-35
- Safety Statements
- 17-27-36/37/39-8-45-26
- RIDADR
- UN 2466 5.1/PG 1
- WGK Germany
- 3
- RTECS
- TT6053000
- TSCA
- Yes
- HazardClass
- 5.1
- PackingGroup
- I
- HS Code
- 28259080
Use
Potassium superoxide is a powerful oxidizer. Forms on the surface of potassium metal, solid or molten, that is exposed to the air. Attempts to extinguish burning potassium with powdered graphite has resulted in violent explosions [Chem. Abstr. 63:424. 1965]. Highly oxidized potassium metal was dropped into a dish of ethyl alcohol, an immediate explosion shattered the dish. Potassium superoxide was considered the cause of the reaction [Health and Safety Inf. 251. 1967]. Potassium superoxide should not be added to pure organic materials (hydrocarbons), as ignition and violent explosion may occur. Oxidation of arsenic, antimony, copper, potassium, tin, or zinc proceeds with incandescence, [Mellor, 1941, Vol. 2, 493]. Interaction between the superoxide and diselenium dichloride is violent, [Mellor, 1947, Vol. 10, 897].
According to relevant laws, regulations, policies and the provisions of this website, this website does not display the product information!
The source is "易制爆、炸药、化学武器等". If the information is inaccurate, please contact the platform to modify it.
contact information:info@chemicalbook.com 400-158-660