Ceric Sulphate manufacturers
|
| | Ceric Sulphate Basic information |
| Product Name: | Ceric Sulphate | | Synonyms: | cericdisulfate;Cericsulfate,anhydrous;Sulfuricacid,cerium(4+)salt(2:1);sulfuricacid,cerium(4++)salt(2:1);sulfuricacid,ceriumsalt(2:1);cerium(iv) sulfate solution;Cerium(IV) sulfate, anhydrous, 98+%;CERIUM(IV) SULFATE, 0.25N SOLUTION IN 1-4N SULFURIC ACID | | CAS: | 13590-82-4 | | MF: | CeH2O4S | | MW: | 238.19 | | EINECS: | 237-029-5 | | Product Categories: | Inorganics | | Mol File: | 13590-82-4.mol | 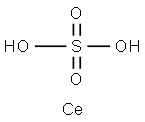 |
| | Ceric Sulphate Chemical Properties |
| | Ceric Sulphate Usage And Synthesis |
| Description | Ceric sulphate salts are yellow to yellow/orange solids that are moderately soluble in water and dilute acids. Its neutral solutions slowly decompose, depositing the light yellow oxide CeO2.
| | Physical properties |
Ceric Sulphate is a white crystalline powder with orthogonal crystal system, insoluble in glacial acetic acid and pure ethanol(96%).
| | Uses |
Ceric sulfate is used in analytical chemistry for redox titration, often together with a redox indicator. It is also used to prepare spherical-like nanoparticles CeO2, nanoparticles for NO reductions' reactions.
| | Preparation | Ceric Sulphate was historically produced by direct reaction of fine, calcined cerium (IV) oxide and concentrated sulfuric acid, yielding the tetrahydrate.
| | Health Hazard | Experimental reproductive effects. When heated to decomposition it emits acrid smoke and irritating fumes.
|
| | Ceric Sulphate Preparation Products And Raw materials |
|