|
|
| | Cerium Nitrate Hexahydrate Basic information | | Uses |
| Product Name: | Cerium Nitrate Hexahydrate | | Synonyms: | Cerium(III)nitrate,hexahydrate(1:3:6);Ceriumtrinitratehexahydrate;Nitricacid,cerium(3+)salt,hexahydrate;cerium(iii) nitrate hexahydrate, reacton;CERIUM(III)NITRATE,CRYSTALLINE;CERIUM(III)NITRATE,REAGENT;CERIUM(III) NITRATE HEXAHYDRATE: (99.99% CE) (REO);Cerium(III) nitrate hexahydrate, REacton(R), 99.99% (REO) | | CAS: | 10294-41-4 | | MF: | CeH3NO4 | | MW: | 221.14 | | EINECS: | 600-370-9 | | Product Categories: | Rare earth;metal nitrate salts | | Mol File: | 10294-41-4.mol | 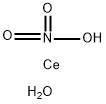 |
| | Cerium Nitrate Hexahydrate Chemical Properties |
| Melting point | 96℃ | | Boiling point | 200°C | | density | 4.37 | | vapor density | 15 (vs air) | | storage temp. | Store at +15°C to +25°C. | | solubility | 1754g/l | | form | Crystalline Powder or Agglomerating Crystals | | color | Colorless to white | | PH | 3.7 (100g/l, H2O, 25℃) | | Water Solubility | Soluble in water, alcohol and acetone. | | Sensitive | Hygroscopic | | Decomposition | >200°C | | Merck | 14,2000 | | Stability: | Stable. Hygroscopic and air sensitive. Oxidizing. Incompatible with strong reducing agents, strong bases. | | CAS DataBase Reference | 10294-41-4(CAS DataBase Reference) | | EPA Substance Registry System | Cerium(III) nitrate hexahydrate (10294-41-4) |
| Hazard Codes | O,Xi,N | | Risk Statements | 8-41-50/53-52/53-36/38 | | Safety Statements | 17-26-39-61-60 | | RIDADR | UN 1477 5.1/PG 2 | | WGK Germany | 2 | | RTECS | FK6300000 | | F | 3-10-23 | | TSCA | Yes | | HazardClass | 5.1 | | PackingGroup | III | | HS Code | 28461090 | | Toxicity | LD50 orally in Rabbit: 4200 mg/kg |
| | Cerium Nitrate Hexahydrate Usage And Synthesis |
| Chemical Properties | Cerium nitrate hexahydrate is a widely used source of cerium in materials science. It is a white-to-yellow crystalline salt that is hygroscopic and air-sensitive. It is highly soluble in water, alcohol, and acetone, although solutions can appear slightly hazy. Like many trivalent metal nitrates, cerium (III) nitrate hexahydrate melts at a low temperature (57 °C)and thermally decomposes at low temperatures too, beginning at 190 °C and proceeding rapidly at 280 °C with complete decomposition to cerium oxide at 390-400 °C. | | Uses | Cerium (III) nitrate hexahydrate is widely used as a source of cerium, especially in the synthesis of micro- or nano-structured ceria (cerium oxide). Because of its high solubility and low decomposition temperature, cerium (III) nitrate hexahydrate is an ideal reagent for hydrothermal reactions, sol-gel processing, and co-precipitation and calcination reactions. It is also applicable in studies using cerium (III)nitrate hexahydrate to build a ceria support for single-atom catalysts, such as nickel-doped ceria for the hydrogenation of acetylene and platinum-doped ceria for carbon monoxide oxidation. |
| | Cerium Nitrate Hexahydrate Preparation Products And Raw materials |
|