| Company Name: |
|
| Tel: |
|
| Email: |
export1@cleanchemlab.com |
| Products Intro: |
Cas:22818-40-2
ProductName:Amoxicilline Impurity-I(4-Hydroxyphenylglycine)
|
- Amoxicilline Trihydrate
-

- $10.00 / 1Kg/Bag
-
2021-10-30
- CAS:61336-70-7
- Min. Order: 1Kg/Bag
- Purity: 99%
- Supply Ability: 20 Tons
|
| | Amoxicilline Chemical Properties |
| | Amoxicilline Usage And Synthesis |
| Brand Name(s) | Amoxicillin Amoxi-Tabs, Amoxi-Drops, Amoxi-Inject, Robamox-V, Biomox, and other brands; Amoxil, Trimox, Wymox, Polymox, (human forms); Amoxicillin trihydrate
| | Chemical Properties | solid | | Originator | Arnoxil,Bencard,UK,1972 | | Uses | Amoxicillin is an antibiotic used in the treatment of various bacterial infections. | | Uses | Antibacterial;Bacterial transpeptidase inhibitor | | Definition | ChEBI: A penicillin in which the substituent at position 6 of the penam ring is a 2-amino-2-(4-hydroxyphenyl)acetamido group. | | Indications | Amoxycillin, like ampicillin, has a broad spectrum of use.
Indications for use are the same as with ampicillin. Synonyms of this drug are amoxi�can, amoxil, larotid, robamox, trimox, vimox, utimox, and others.
Undoubtedly, analogs of ampicillin that are substituted at the amine fragment of phenyl�glycine (azolcillin, mezlocillin, piperacillin) should be included in this same group of com�pounds. | | Manufacturing Process | Ethyl chlorocarbonate (2.2 ml) was added to an ice cold solution of O,N-dibenzyloxycarbonyl-p-oxy-dl-α-aminophenylacetic acid (10 grams) and
triethylamine (3.85 ml) in dry acetone (193 ml). The mixture was stirred at
0°C for 5 minutes during which triethylamine hydrochloride precipitated. The
suspension was cooled to -30°C and stirred vigorously while adding as rapidly
as possible an ice cold solution of 6-aminopenicillanic acid (5.85 grams) in 3%
aqueous sodium bicarbonate (193 ml), the temperature of the mixture never
being allowed to rise above 0°C. The resulting clear solution was stirred for 30
minutes at 0°C, and then for a further 30 minutes, without external cooling,
and finally extracted with diethyl ether (3 x 200 ml) only the aqueous phase
being retained.
This aqueous solution was brought to pH 2 by the addition of hydrochloric acid
and the 6-(O,N-dibenzyloxycarbonyl-p-oxy-dl-α-
aminophenylacetamido)penicillanic acid so liberated was extracted into diethyl
ether (50 ml and 2 portions of 30 ml). The ether phase was washed with
water (3 x 5 ml) and the water washings were discarded.
Finally, the penicillin was converted to the sodium salt by shaking the ether
solution with sufficient 3% sodium bicarbonate to give a neutral aqueous
phase, separating the latter and evaporating it at low pressure and
temperature below 20°C. The product was finally dried over phosphorus
pentoxide in vacuo to give sodium 6-(O,N-dibenzyloxycarbonyl-p-oxy-dl-α-
aminophenylacetamido)-penicillanate (9.2 grams).
A suspension of palladium on calcium carbonate (36 grams of 5%) in water
(150 ml) was shaken in an atmosphere of hydrogen at room temperature and
atmospheric pressure for 1 hour. A neutral solution of sodium 6-(O,Ndibenzyloxycarbonyl-
p-oxy-dl-α-aminophenylacetamido)penicillanate (9
grams) in water (100 ml) was then added and shaking in hydrogen was
resumed for one hour. The suspension was then filtered and the collected
catalyst was washed well with water without being allowed to suck dry
between washings. The combined filtrate and washings were then brought to
pH 6.5 with dilute hydrochloric acid and evaporated to dryness at reduced
pressure and temperatures below 20°C. The product was finally dried over
phosphorus pentoxide in vacuo to give a solid (5.4 grams) containing 6-(phydroxy-
dl-α-aminophenylacetamido)penicillanic acid. | | Brand name | Amoxil (GlaxoSmithKline);
Dispermox (Ranbaxy); Larotid (GlaxoSmithKline);
Trimox (Apothecon) [Name previously used:
Amoxycillin.]. | | Therapeutic Function | Antibacterial | | Antimicrobial activity | The antibacterial spectrum is identical to that of ampicillin
and there are few differences in antibacterial activity
. Like ampicillin, amoxicillin is unstable to most
β-lactamases. It has useful activity against Helicobacter pylori
(<1% resistance), and is included in most combination regimens
for the treatment of H. pylori infections. | | Acquired resistance | There is complete cross-resistance with ampicillin. Its
action against many β-lactamase-producing strains can be
restored by co-administration with β-lactamase inhibitors. | | Flammability and Explosibility | Non flammable | | Biochem/physiol Actions | Amoxicillin fights infections in the body within an hour after taking it. The penicillin-based medication gets its strength from consistent use of multiple doses per day. Amoxicillin often relieves symptoms in less than 72 hours and stays in your system for 24 hours. | | Contact allergens | Amoxicillin is both a topical and a systemic sensitizer.
Topical sensitization occurs in health care workers.
Systemic drug reactions are frequent, such as urticaria,
maculo-papular rashes, baboon syndrome, acute generalized
exanthematous pustulosis, or even toxic epidermal
necrosis. Cross-reactivity is common with
ampicillin, and can occur with other penicillins. | | Mechanism of action | Amoxicillin shows a bactericidal (kills microorganisms) effect against susceptible organisms (bacteria that are unable to grow in the presence of the drug) during their stage of active multiplication. Amoxicillin mode of action is similar to ampicillin, and thus it works by preventing the synthesis of the mucopeptide (a protein responsible for the growth of bacteria) present in the cell wall, which in turn leads to the death of the bacteria. | | Pharmacokinetics | Oral absorption: 75–90%
Cmax 500 mg oral: 5.5–7.6 mg/L after 1–2 h
500 mg intramuscular: c. 14 mg/L after 1–2 h
Plasma half-life: 1 h
Volume of distribution: 0.3 L/kg
Plasma protein binding: 17–20%
Absorption and distribution
Oral absorption produces over twice the peak concentration
achieved by comparable doses of ampicillin, allowing
less frequent dosing intervals. Absorption is unaffected by
food.
It is well-distributed in multiple body fluids, including
pleural, peritoneal and middle ear fluid. It does not penetrate
well into the CSF.
Metabolism and excretion
Some 10–25% is converted to the penicilloic acid.
Between 50% and 70% of unchanged drug is recovered
in the urine in the first 6 h after a dose of 250 mg. Plasma
levels are elevated and prolonged by the administration of
probenecid. | | Clinical Use | Amoxicillin, 6-[D-(-)-α-amino-p- hydroxyphenylacetamido]penicillanic acid (Amoxil, Larotid, Polymox), a semisyntheticpenicillin introduced in 1974, is simply the p-hydroxyanalog of ampicillin, prepared by acylation of 6-APA with phydroxyphenylglycine.Its antibacterial spectrum is nearly identical with that ofampicillin, and like ampicillin, it is resistant to acid, susceptibleto alkaline and β-lactamase hydrolysis, andweakly protein bound. Early clinical reports indicated thatorally administered amoxicillin possesses significantadvantages over ampicillin, including more complete GIabsorption to give higher plasma and urine levels, lessdiarrhea, and little or no effect of food on absorption.50Thus, amoxicillin has largely replaced ampicillin for thetreatment of certain systemic and urinary tract infectionsfor which oral administration is desirable. Amoxicillin isreportedly less effective than ampicillin in the treatment ofbacillary dysentery, presumably because of its greater GIabsorption. Considerable evidence suggests that oral absorptionof α-aminobenzyl–substituted penicillins (e.g.,ampicillin and amoxicillin) and cephalosporins is, at leastin part, carrier mediated, thus explaining their generallysuperior oral activity.Amoxicillin is a fine, white to off-white, crystallinepowder that is sparingly soluble in water. It is available invarious oral dosage forms. Aqueous suspensions are stablefor 1 week at room temperature. | | Clinical Use | Isolates should be tested for susceptibility before use, especially
for serious infections.
Ear, nose and throat infections other than pharyngitis, which may mask
glandular
fever
Tracheobronchitis, bronchitis, pneumonia
Genitourinary tract infections, including gonorrhea
Infections of skin and soft tissues due to streptococci and susceptible
staphylococci
Helicobacter pylori infection (in combination with a proton pump inhibitor
and at least one other antimicrobial agent such as clarithromycin)
Prophylaxis of endocarditis in patients undergoing dental treatment and
other procedures | | Side effects | Amoxicillin is generally well tolerated, side effects being those
common to penicillins, but including non-allergic rashes in
patients with glandular fever. As the drug is well absorbed,
diarrhea is generally infrequent and rarely sufficiently severe
to require withdrawal of treatment.
Common side effects of Amoxicillin include Diarrhoea (loose stools), Headache, Nausea (vomiting sensation), Vomiting, Rash, Anaphylaxis (allergic condition), and Anaemia (lack of blood).
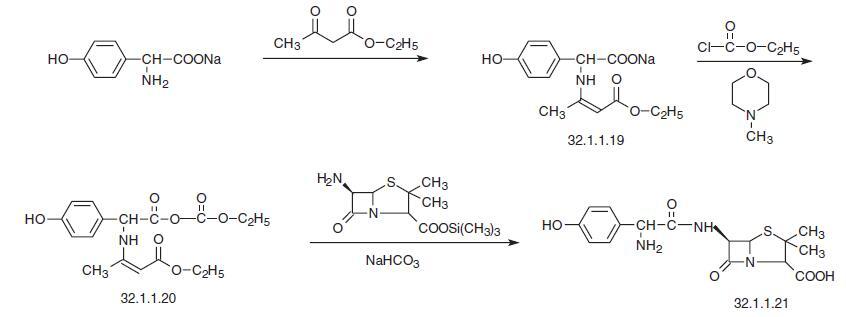
Serious side effects of Amoxicillin include Thrombocytopenia (deficiency of platelets), Convulsions (involuntary movements of body muscles), Cholestatic jaundice (jaundice caused due to the stoppage of bile from the liver), Meningitis (inflammation of brain and spinal membranes), and Vasculitis (inflammation of blood vessels). | | Synthesis | Amoxycillin, [2S-[2α,5α,6β(S)]]-3,3-dimethyl-7-6-[[amino-(4-hydroxyph�enyl)-acetyl]amino]-4-thia-1-azabicyclo[3.2.0]-heptan-2-carboxylic acid (32.1.1.21), is syn�thesized in two ways. The first uses an enamine protection of the amino group of
4-hydroxyphenylglycine, which begins with the sodium salt of 4-hydroxyphenylglycine,
which is reacted with the acetoacetic ester to form an enamine—the sodium salt of a p�hydroxyphenyl acetic acid, α-[(3-ethoxy-1-methyl-3-oxo-1-propenyl)amino]-4-hydroxy-
(32.1.1.19). Reacting the resulting aminocrotonate with the ethyl chloroformate in
N-methylmorpholine gives the corresponding mixed anhydride (32.1.1.20), which is reacted
with trimethylsilyl ester of 6-APA.
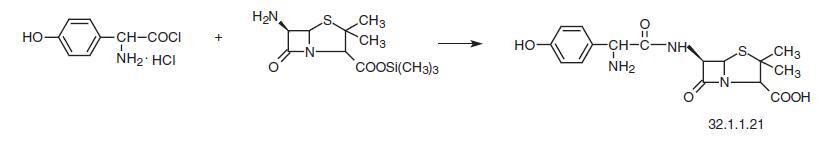
The second method uses a direct reaction of D-(-)-2-(4-hydroxyphenyl)glycine chloride
hydrochloride with trimethylsylil ester of 6-APA.
The trimethylsilyl ester of 6-APA needed for the reaction is in turn synthesized by react�ing trimethylchlorosilylane with 6-APA in the presence of trimethylamine. | | Veterinary Drugs and Treatments | The aminopenicillins have been used for a wide range of infections
in various species. FDA-approved indications/species, as well as
non-approved uses, are listed in the Dosages section below. | | Veterinary Drugs and Treatments | Amoxicillin/potassium clavulanate tablets and oral suspension
products are approved
for use in dogs and cats for the treatment of
urinary tract, skin and soft tissue infections caused by susceptible
organisms. It is also indicated for canine periodontal disease due to
susceptible strains of bacteria. | | Drug interactions | Potentially hazardous interactions with other drugs
Amoxicillin can reduce the excretion of methotrexate
(increased risk of toxicity). | | Metabolism | Amoxicillin is metabolised to a limited extent to
penicilloic acid which is excreted in the urine. About 60%
of an oral dose of amoxicillin is excreted unchanged in
the urine by glomerular filtration and tubular secretion.
Probenecid reduces renal excretion. High concentrations
have been reported in bile; some may be excreted in the
faeces. |
| | Amoxicilline Preparation Products And Raw materials |
|