|
|
| | Pimodivir Basic information |
| Product Name: | Pimodivir | | Synonyms: | Pimodivir;Pimodivir (VX-787);VX-787;VRT0928787;VRT-0928787;VX-787; VX 787; VX787; JNJ-872; JNJ 872; JNJ872; VRT-0928787; VRT 0928787; VRT0928787;PIMODIVIR;VX-787 (Pimodivir);Bicyclo[2.2.2]octane-2-carboxylic acid, 3-[[5-fluoro-2-(5-fluoro-1H-pyrrolo[2,3-b]pyridin-3-yl)-4-pyrimidinyl]amino]-, (2S,3S)- | | CAS: | 1629869-44-8 | | MF: | C20H19F2N5O2 | | MW: | 399.39 | | EINECS: | | | Product Categories: | | | Mol File: | 1629869-44-8.mol | 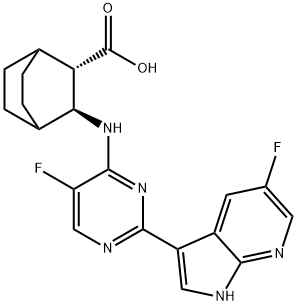 |
| | Pimodivir Chemical Properties |
| density | 1.501±0.06 g/cm3(Predicted) | | storage temp. | Store at -20°C | | solubility | Soluble in DMSO, not in water:100.0(Max Conc. mg/mL);250.38(Max Conc. mM) | | form | A solid | | pka | 4.32±0.40(Predicted) | | color | White to light yellow |
| | Pimodivir Usage And Synthesis |
| Description | Pimodivir is an inhibitor of influenza virus polymerase basic protein 2 (PB2; KD = <0.003 μM). It also binds to glycogen synthase kinase 3β (GSK3β; Ki = ~1.6 μM) and inhibits the activity of Axl and calcium/calmodulin-dependent protein kinase IIβ (CaMKIIβ) by greater than 50% in a panel of 65 human and rat kinases. Pimodivir decreases the replication of seven adamantine- and neuraminidase inhibitor-resistant strains of influenza virus A (EC50s = <0.15-2.8 nM in a cell-based assay). It increases the antiviral activity of oseltamivir , zanamivir , and favipiravir (T-705; ) with 50% combination index (CI50) values of 0.58, 0.64, and 0.89, respectively, in a cell-based assay. Pimodivir increases survival in a mouse model of intranasal influenza A infection when administered at doses of 1, 3, and 10 mg/kg twice per day. | | Uses | Pimodivir is an orally bioavailable azaindole inhibitor of influenza PB2. |
| | Pimodivir Preparation Products And Raw materials |
|