- Cupric tartrate
-

- $15.00 / 1KG
-
2021-08-12
- CAS:815-82-7
- Min. Order: 1KG
- Purity: 99%+ HPLC
- Supply Ability: Monthly supply of 1 ton
- Cupric Tartrate
-

- $0.00 / 1KG
-
2020-02-26
- CAS: 815-82-7
- Min. Order: 1KG
- Purity: 99.0%+
- Supply Ability: 100 tons
- Cupric tartrate
-

- $1.00 / 1kg
-
2019-07-06
- CAS:815-82-7
- Min. Order: 1kg
- Purity: 98%min
- Supply Ability: as request
|
| | Cupric tartrate Basic information |
| Product Name: | Cupric tartrate | | Synonyms: | TARTARIC ACID CUPRIC SALT TRIHYDRATE;2,3-dihydroxy-(r-(r*,r*))-butanedioicacicopper(2+)salt(1:1);2,3-dihydroxy-[theta-(theta,theta)]-butanedioicacicopper(2++)salt(1;CUPRIC TARTRATE;CUPRIC TARTRATE HYDRATE;CUPRIC TARTRATE TRIHYDRATE;COPPER TARTRATE TRIHYDRATE;COPPER(II) TARTRATE | | CAS: | 815-82-7 | | MF: | C4H6CuO6 | | MW: | 213.63 | | EINECS: | 212-425-0 | | Product Categories: | salt of an organic acid;Organic-metal salt | | Mol File: | 815-82-7.mol | 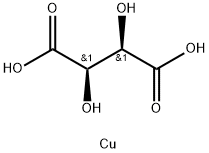 |
| | Cupric tartrate Chemical Properties |
| Melting point | ~275 °C (dec.)(lit.) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | solubility | slightly soluble in H2O; soluble in acid solutions, alkaline solutions | | form | crystal | | color | green to blue | | Water Solubility | slightly soluble | | CAS DataBase Reference | 815-82-7(CAS DataBase Reference) | | EPA Substance Registry System | Cupric tartrate (815-82-7) |
| | Cupric tartrate Usage And Synthesis |
| Chemical Properties | bluish green crystal(s) [STR93] | | Uses | Copper(II) Tartrate can be useful in gas-induced formation of Cu nanoparticle as catalyst for high-purity straight and helical carbon nanofibers. | | General Description | A green to blue odorless powder. Insoluble in water. The primary hazard is the threat to the environment. Immediate steps should be taken to limit its spread to the environment. Cupric tartrate is noncombustible. Used for electroplating metals. | | Air & Water Reactions | Insoluble in water. | | Reactivity Profile | Acidic salts, such as Cupric tartrate , are generally soluble in water. The resulting solutions contain moderate concentrations of hydrogen ions and have pH's of less than 7.0. They react as acids to neutralize bases. These neutralizations generate heat, but less or far less than is generated by neutralization of inorganic acids, inorganic oxoacids, and carboxylic acid. They usually do not react as either oxidizing agents or reducing agents but such behavior is not impossible. Many of these compounds catalyze organic reactions. | | Health Hazard | INHALATION: Inhalation of dust may cause nasal congestion. EYES: May cause conjunctivitis and edema of eyelids. SKIN: May irritate skin. INGESTION: Vomiting is caused by local irritant and astringent action of ionic copper on stomach and bowel. |
| | Cupric tartrate Preparation Products And Raw materials |
|