|
|
| | Ethyl 8-bromooctanoate Basic information |
| Product Name: | Ethyl 8-bromooctanoate | | Synonyms: | ETHYL W-BROMOOCTANOATE;ETHYL OMEGA-BROMOOCTANOATE;8-BROMOOCTANOIC ACID ETHYL ESTER;8-CAPRYLIC ACID ETHYL ESTER;8-Bromooctanoic Ethylester;Ethyl 8-bromoocatanoate;8-BroMooCLanoic Acid Ethyl Ester;Ethyl 8-broMooctanoate, 98%+ | | CAS: | 29823-21-0 | | MF: | C10H19BrO2 | | MW: | 251.16 | | EINECS: | 608-417-5 | | Product Categories: | 1 | | Mol File: | 29823-21-0.mol | 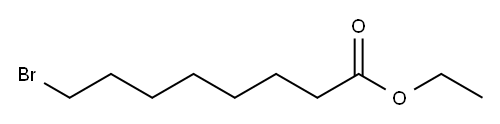 |
| | Ethyl 8-bromooctanoate Chemical Properties |
| Boiling point | 123°C/ 3mm | | density | 1.194 | | storage temp. | 2-8°C | | solubility | DMSO (Slightly), Ethyl Acetate (Slightly) | | form | Oil | | color | Clear Colourless | | InChI | InChI=1S/C10H19BrO2/c1-2-13-10(12)8-6-4-3-5-7-9-11/h2-9H2,1H3 | | InChIKey | UBTQVPMVWAEGAC-UHFFFAOYSA-N | | SMILES | C(OCC)(=O)CCCCCCCBr | | LogP | 3.809 |
| Hazard Codes | Xi | | HazardClass | IRRITANT | | HS Code | 2915907098 |
| | Ethyl 8-bromooctanoate Usage And Synthesis |
| Uses | Ethyl 8-bromooctanoate is a useful research reagent for organic synthesis and other chemical processes. | | Synthesis | The synthesis method of ethyl 8-bromooctanoate: 1, 6-dibromohexane initial raw material is firstly used for carrying out substitution reaction with diethyl malonate to generate 2- (6-bromohexyl) diethyl malonate, and then ester hydrolysis and decarboxylation reaction are carried out to obtain the 8-bromooctanoic acid; and finally, esterification reaction is carried out to generate the compound 8-bromoethyl octanoate. |
| | Ethyl 8-bromooctanoate Preparation Products And Raw materials |
|