|
|
| | 4-ChloroMethyl-1-cyclopentyl-2-trifluoroMethyl-benzene Basic information |
| | 4-ChloroMethyl-1-cyclopentyl-2-trifluoroMethyl-benzene Chemical Properties |
| Boiling point | 293.7±35.0 °C(Predicted) | | density | 1.226±0.06 g/cm3(Predicted) | | InChI | InChI=1S/C13H14ClF3/c14-8-9-5-6-11(10-3-1-2-4-10)12(7-9)13(15,16)17/h5-7,10H,1-4,8H2 | | InChIKey | NUFXNFBJDGCADP-UHFFFAOYSA-N | | SMILES | C1(C2CCCC2)=CC=C(CCl)C=C1C(F)(F)F |
| | 4-ChloroMethyl-1-cyclopentyl-2-trifluoroMethyl-benzene Usage And Synthesis |
| Uses |
4-chloromethyl-1-cyclopentyl-2-(trifluoromethyl)benzene is a precursor of APD334, which is a potent functional antagonist of S1P1 and has a favourable PK/PD profile, producing robust lymphocyte lowering at relatively low plasma concentrations in several preclinical species[1].
| | Synthesis |
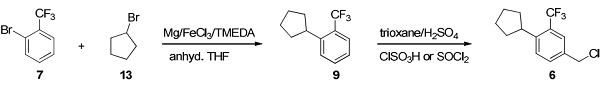
A two-step scalable process was developed starting from commercially available and inexpensive starting materials. An iron(III) chloride-catalyzed aryl–alkyl cross-coupling reaction provided the intermediate 1-cyclopentyl-2-(trifluoromethyl)benzene (9), which was converted to the target building block 4-chloromethyl-1-cyclopentyl-2-(trifluoromethyl)benzene by a direct regioselective chloromethylation reaction with trioxane/thionyl chloride or chlorosulfonic acid in sulfuric acid[2].
| | References |
[1] Daniel J. Buzard*. “Discovery of APD334: Design of a Clinical Stage Functional Antagonist of the Sphingosine-1-phosphate-1 Receptor.” ACS Medicinal Chemistry Letters 5 12 (2014): 1313–1317.
[2] Dipanjan Sengupta. “An Efficient Scale-Up Process for the Preparation of the APD334 Precursor 4-Chloromethyl-1-cyclopentyl-2-(trifluoromethyl)benzene.” Organic Process Research Development 19 6 (2015): 618–623.
|
| | 4-ChloroMethyl-1-cyclopentyl-2-trifluoroMethyl-benzene Preparation Products And Raw materials |
|