- Cis-3-Hexenyl Acetate
-

- $10.00 / 1kg
-
2024-07-25
- CAS:3681-71-8
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 10 ton
- cis-3-Hexenyl Acetate
-

- $0.00 / 25Kg/Drum
-
2024-07-25
- CAS:3681-71-8
- Min. Order: 1KG
- Purity: 98%min
- Supply Ability: 500kg
- cis-3-Hexenyl Acetate
-

- $0.00 / 1kg
-
2024-07-21
- CAS:3681-71-8
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10ton
|
| | cis-3-Hexenyl Acetate Basic information |
| Product Name: | cis-3-Hexenyl Acetate | | Synonyms: | Zinc05225119;cis-3-Hexenyl acetate,(3Z)-C-3-Hexenyl acetate, 3-Hexen-1-ol, Leaf acetate;VERDURAL;CIS-3-HEXENYL ACETATE 98+% FCC;Leaf Acetate or CIS-3-Hexenyl Acetate 98%;CIS-3-Hexenyl Acetate 98%;Leaf acetate
cis-3-Hexenyl acetate;Z-3-hexenyl-1-acetate | | CAS: | 3681-71-8 | | MF: | C8H14O2 | | MW: | 142.2 | | EINECS: | 222-960-1 | | Product Categories: | | | Mol File: | 3681-71-8.mol | 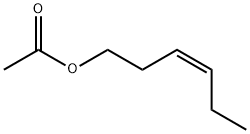 |
| | cis-3-Hexenyl Acetate Chemical Properties |
| Risk Statements | 36/38-10 | | Safety Statements | 26-36-24/25 | | RIDADR | UN 3272 3/PG 3 | | WGK Germany | 2 | | RTECS | MP8425050 | | TSCA | Yes | | HazardClass | 3 | | PackingGroup | III | | HS Code | 29153900 | | toxicity | Both the acute oral LD50 value in
rats and the acute dermal LD50 value in rabbits exceeded 5 g/kg (Wohl, 1974). |
| | cis-3-Hexenyl Acetate Usage And Synthesis |
| Description | cis-3-Hexen-1 -y 1-acetate has a powerful green, floral note reminiscent of banana. It may be prepared by acetylation of the corresponding alcohol. | | Chemical Properties | Colorless liquid | | Chemical Properties | (Z)-3-Hexenyl Acetate has been identified in many fruit aromas and
green tea. It is a prototype for green odors and is often used in combination with
(Z)-3-hexenol. | | Chemical Properties | cis-3-Hexen-1-yl acetate has a powerful, green, fruity, floral note reminiscent of banana. | | Occurrence | Reported found in apple, bilberry, guava, strawberry, black and green tea, yellow and purple passion fruit,
citrus peel oils, melon, peach, raspberry, pear, pineapple, melon, celery, tomato, corn mint oil, spearmint oil, brandies, grape wines,
soybeans, olive, plum, plumcot, starfruit, mango, cauliflower, dill, lovage, corn oil, nectarines, Chinese quince and chamomile oil. | | Uses | Leaf Acetate increased leaf-feeding herbivores’ egg predation rates by a generalist predator. As a consequence, a plant could reduce the number of herbivores by more than 90% by releasing volatiles. | | Preparation | By acetylation of the corresponding alcohol | | Definition |
ChEBI: An acetate ester that results from the formal condensation of acetic acid with (Z)-hex-3-en-1-ol.
In dilutions below 10 ppm, it produces very pleasant fruity-green flavours, and its taste individually at concentrations below 2 ppm is distinctly Banana-1ike. Very interesting combination flavors can be created with this ester and a few other chemicals: A concentration of about 1 ppm of cis-3-Hexenyl acetate in the presence of 0.05 ppm Allyl-iso-thiocyanate (Allyl mustard oil) will produce a distinct note of unripe Banana with the sharp greenness and the fruity notes unmistakably identifiable as Banana and absolutely no impression of Mustard. The two materials separately at the concentrations indicated give very weak and incomplete flavor pictures. However, this ester finds considerable use in perfumes, where it may lend pleasant and natural freshness to delicate florals (Muguet, Lilac, Narcisse, etc.) and participate in creating true-to-nature, foliage-green topnotes in other fragrance types. The ester is most likely to supersede the less stable and less natural-smelling Alkyl acetylenic esters, slowly being abandoned in modern perfumery.
| | Taste threshold values | Taste characteristics at 10 ppm: green, fruity, apple and pear with fresh tropical nuances | | Synthesis Reference(s) | Canadian Journal of Chemistry, 65, p. 2327, 1987 DOI: 10.1139/v87-388 | | Flammability and Explosibility | Flammable |
| | cis-3-Hexenyl Acetate Preparation Products And Raw materials |
|