|
|
| | Pipobroman Basic information |
| Product Name: | Pipobroman | | Synonyms: | 1,1'-(Piperazine-1,4-diyl)bis(3-bromopropan-1-one);Pipobroman;1,4-Bis(3-bromo-1-oxopropyl)piperadine;A-8103;Amedel;NSC-25154;Vercyte;3-bromo-1-[4-(3-bromopropanoyl)piperazin-1-yl]propan-1-one | | CAS: | 54-91-1 | | MF: | C10H16Br2N2O2 | | MW: | 356.05 | | EINECS: | | | Product Categories: | HYDROMOX | | Mol File: | 54-91-1.mol | 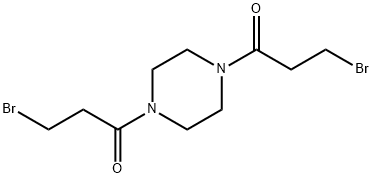 |
| | Pipobroman Chemical Properties |
| Melting point | 106-107℃ | | Boiling point | 487℃ | | density | 1.691 | | refractive index | 1.6220 (estimate) | | Fp | 248℃ | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | solubility | Chloroform (Slightly), Methanol (Slightly, Heated) | | form | Solid | | pka | -0.56±0.70(Predicted) | | color | Crystals from H2O | | EPA Substance Registry System | Pipobroman (54-91-1) |
| | Pipobroman Usage And Synthesis |
| Originator | Vercyte ,Abbott ,US ,1966 | | Uses | diuretic, antihypertensive | | Definition | ChEBI: Pipobroman is an N-acylpiperazine that is piperazine in which each of the nitrogens has been acylated by a 3-bromopropionoyl group. An anti-cancer drug. It has a role as an antineoplastic agent and an alkylating agent. It is a N-acylpiperazine, a tertiary carboxamide and an organobromine compound. | | Manufacturing Process | To a solution of 17.2 g (0.10 mol) of 3-bromopropionyl chloride in 100 ml of anhydrous benzene was added dropwise with stirring a solution of 8.6 g (0.10 mol) of anhydrous piperazine in 20 ml of dry chloroform over a period of 30 minutes. The temperature rose spontaneously to 45°C during the addition. After the temperature ceased to rise, stirring was continued for another hour. The reaction mixture was then filtered to remove the piperazine hydrochloride by-product. The filtrate was evaporated to dryness and the residue recrystallized from ethanol to obtain the desired N,N'-bis-(3bromopropionyl)piperazine as a white crystalline solid melting at 103°C to 104°C. The identity of the product was further established by elemental analysis. | | Brand name | Vercyte (Abbott). | | Therapeutic Function | Antineoplastic | | Safety Profile | Poison by ingestion,subcutaneous and intraperitoneal routes. An experimentalteratogen. Other experimental reproductive effects.Human mutation data reported. When heated todecomposition it emits very toxic fumes of Br- and NOx. |
| | Pipobroman Preparation Products And Raw materials |
|