| Company Name: |
Shanghai Aladdin Bio-Chem Technology Co.,LTD
|
| Tel: |
400-400-6206333 18521732826 |
| Email: |
market@aladdin-e.com |
| Products Intro: |
Product Name:Dibenzotetrathiafulvalene
CAS:24648-13-3
Purity:97% Package:500mg/RMB 727.90;100mg/RMB 739.90;25mg/RMB 299.90
|
|
| | Dibenzotetrathiafulvalene Basic information |
| Product Name: | Dibenzotetrathiafulvalene | | Synonyms: | Δ2,2'-Bi[4,5-[1,3]butadieno-1,3-dithiol];Dibenzotetrathiafulvalene 97%;DBTTF;1,3-Benzodithiole,2-(1,3-benzodithiol-2-ylidene)-;[2,2]Bi[benzo[1,3]dithiolylidene], DB-TTF;Dibenzotetrathiafulvalene;Δ2,2'-Bi(1,3-benzodithiol);Δ2,2'-Bi[1,3-benzodithiole] | | CAS: | 24648-13-3 | | MF: | C14H8S4 | | MW: | 304.47 | | EINECS: | | | Product Categories: | | | Mol File: | 24648-13-3.mol | 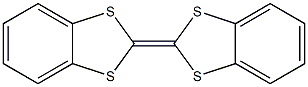 |
| | Dibenzotetrathiafulvalene Chemical Properties |
| Melting point | 239-243 °C | | Boiling point | 367.1±42.0 °C(Predicted) | | density | 1.538±0.06 g/cm3(Predicted) |
| Hazard Codes | N | | Risk Statements | 50/53 | | Safety Statements | 60-61 | | RIDADR | UN 3077 9/PG 3 | | WGK Germany | 3 |
| | Dibenzotetrathiafulvalene Usage And Synthesis |
| Uses | Dibenzotetrathiafulvalene(DBTTF, also known as 1,3-Benzodithiole,2-(1,3-benzodithiol-2-ylidene)-) is an organic electronic material that can be used to form semiconductor charge transfer salts with electron accepting (n-type) materials (TCNQ and F4TCNQ). It can also be used to form charge transfer complexes (TCNQ and DDQ) with a variety of semiconductor crystals for the manufacture of organic field effect transistors (OFETs) and organic light emitting diodes (OLEDs). | | Synthesis | Dibenzotetrathiafulvalene was prepared from 2,2'-Bi-1,3-benzodithiol under reaction conditions with 2,3-dicyano-5,6-dichloro-p-benzoquinone and dichloromethane. The specific method of operation is described below:
General procedure: Dithioacetal (0.1 mmol) and DDQ (68 mg, 0.30 mol) were dissolved in CH2Cl2 (3 mL). The resultant dark blue mixture was stirred at room temperature for 16 h and was then washed with a saturated NaOH aqueous solution (2 mL) and CH2Cl2 (10 mL). The orange organic phase was dried over anhydrous Na2SO4, filtered, and then evaporated under reduced pressure. The crude solid was recrystallized from hexane to give the corresponding dibenzotetrathiafulvalene derivative. Dibenzotetrathiafulvalene. Yield: 30 mg (99%); orange solid; mp 239.8-243.0 °C. 1H NMR (500 MHz, CDCl3): δ = 7.27-7.24 (m, 4 H), 7.13-7.10 (m, 4 H). 13C NMR (125 MHz, CDCl3): δ = 136.6, 125.9, 121.9, 110.6. MS (FAB): m/z = 304 [M+]. HRMS (FAB): m/z calcd for [M]+ (C14H8S4): 303.9509; found: 303.9519.
 |
| | Dibenzotetrathiafulvalene Preparation Products And Raw materials |
|