(R)-(-)-HEXAHYDROMANDELIC ACID manufacturers
|
| | (R)-(-)-HEXAHYDROMANDELIC ACID Basic information |
| Product Name: | (R)-(-)-HEXAHYDROMANDELIC ACID | | Synonyms: | (R)-(-)-HEXAHYDROMANDELIC ACID;R(-)-CYCLOHEXYLHYDROXYACETIC ACID;2-CYCLOHEXYLGLYCOLIC ACID;ALPHA-HYDROXYCYCLOHEXANE-ACETIC ACID;CYCLOHEXYL-HYDROXY-ACETIC ACID;D-HEXAHYDROMANDELIC ACID;Cyclohexylglycolic acid;hexahydromandelic acid | | CAS: | 53585-93-6 | | MF: | C8H14O3 | | MW: | 158.2 | | EINECS: | | | Product Categories: | | | Mol File: | 53585-93-6.mol | 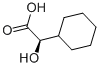 |
| | (R)-(-)-HEXAHYDROMANDELIC ACID Chemical Properties |
| Melting point | 127-129 °C (lit.) | | Boiling point | 319.3±15.0 °C(Predicted) | | density | 1.178±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,Room Temperature | | pka | 3.86±0.11(Predicted) | | optical activity | [α]18/D 23°, c = 1 in acetic acid | | BRN | 3196809 |
| | (R)-(-)-HEXAHYDROMANDELIC ACID Usage And Synthesis |
| Uses | (R)-(?)-Hexahydromandelic acid can be used:
- As an intermediate in the synthesis of antimycobacterial compound pyridomycin analogs.
- As a model α-hydroxy acid compound in the chirality sensing studies of organic probes using circular dichroism technique.
- As a starting material in the synthesis of chiral ionic liquids, which are applicable as chiral solvents in asymmetric synthesis and as chiral stationary phases in chromatographic separations.
| | Purification Methods | It forms hexagonal clusters on recrystallisation from CCl4 or Et2O. [Wood & Comley J Chem Soc 2638 1924, Lettré et al. Chem Ber 69 1594 1936]. The racemate has m 137.2-137.6o (134-135o) [Smith et al. J Am Chem Soc 71 3772 1949]. [Beilstein R-10 II 5; S-10 II 6.] |
| | (R)-(-)-HEXAHYDROMANDELIC ACID Preparation Products And Raw materials |
|