- LIXIVAPTAN
-

- $36.00 / 1mg
-
2024-11-19
- CAS:168079-32-1
- Min. Order:
- Purity: 98.36%
- Supply Ability: 10g
- LIXIVAPTAN
-

- $1.00 / 1KG
-
2019-12-26
- CAS:168079-32-1
- Min. Order: 1KG
- Purity: 97%-99.9%
- Supply Ability: 100kg
|
| | LIXIVAPTAN Basic information |
| Product Name: | LIXIVAPTAN | | Synonyms: | LIXIVAPTAN;N-[3-Chloro-4-(5H-pyrrolo[2,1-c][1,4]benzodiazepin-10(11H)-ylcarbonyl)phenyl]-5-fluoro-2-methylbenzamide;VPA 985;BenzaMide,N-[3-chloro-4-(5H-pyrrolo[2,1-c][1,4]benzodiazepin-10(11H)-ylcarbonyl)phenyl]-5-fluoro-2-Methyl-;N-[3-Chloro-4-(5H-pyrrolo[2,1-c][1,4]benzodiazepine-10(11H)-ylcarbonyl)phenyl]-5-fluoro-2-methylbenzamide;N-[3-chloro-4-(6,11-dihydropyrrolo[2,1-c][1,4]benzodiazepine-5-carbonyl)phenyl]-5-fluoro-2-methyl-benzamide;Lixivaptan (VPA-985);VPA985;LIXIVAPTAN (VPA 985) | | CAS: | 168079-32-1 | | MF: | C27H21ClFN3O2 | | MW: | 473.93 | | EINECS: | | | Product Categories: | Aromatics;Heterocycles;Intermediates & Fine Chemicals;Pharmaceuticals | | Mol File: | 168079-32-1.mol | 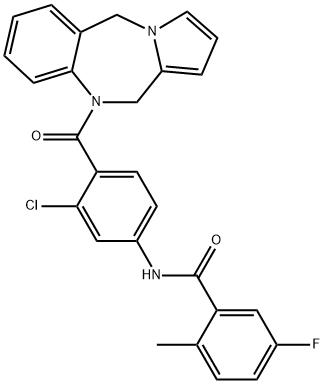 |
| | LIXIVAPTAN Chemical Properties |
| Melting point | >114°C (dec.) | | Boiling point | 626.5±55.0 °C(Predicted) | | density | 1.32 | | storage temp. | Sealed in dry,Room Temperature | | solubility | DMSO: soluble20mg/mL, clear | | form | powder | | pka | 11.86±0.70(Predicted) | | color | white to beige | | InChIKey | PPHTXRNHTVLQED-UHFFFAOYSA-N |
| | LIXIVAPTAN Usage And Synthesis |
| Uses | Lixivaptan is a drug used in the treatment and prevention of cardiovascular diseases. | | Biological Activity | Arginine vasopressin (AVP) plays an important part in circulatory and water homoeostasis and is important in renal hemodynamic alterations, water retention, and cardiac remodeling in congestive heart failure (CHF). Lixivaptan is an AVP V2-receptor selective antagonist th at increases water excretion and normalizes serum sodium levels. Lixivaptan has been in clinical trials for hyponatremia associated with heart failure. | | in vivo | In conscious dogs, water-loaded with 30 mL/kg (po) and arginine vasopressin (AVP)-treated (0.4 μg/kg in oil, sc), lixivaptan (1, 3, and 10 mg/kg po) increases Uvol over the AVP-treated vehicle group by 438, 1018, and 1133%, respectively, while Uosm decreases from 1222 mOsm/kg (water-loaded and AVP treated vehicle) to 307, 221, and 175 mOsm/kg, respectively. In homozygous Brattleboro rats lacking AVP, lixivaptan at 10 mg/kg po (i.e., 10 times the dose producing V2 antagonist activity) b.i.d. for 5 days, shows a sustained antagonist action without evidence of agonist effects. In a randomized double-blind placebo-controlled ascending single dose study, patients (deprived of fluids overnight before dosing) are dosed orally with 30, 75, or 150 mg of lixivaptan. All three doses increase urine flow and serum sodium concentrations and produced significant dose-related decreases in urinary osmolality[1]. Phase II clinical trials in patients with congestive heart failure, liver cirrhosis with ascites or syndrome of inappropriate antidiuretic hormone have demonstrated that lixivaptan increases water clearance without affecting renal sodium excretion or activating the neurohormonal system[2]. | | IC 50 | V2 Receptor |
| | LIXIVAPTAN Preparation Products And Raw materials |
|