|
|
| | BIS- BOC-SPERMIDINE Basic information |
| Product Name: | BIS- BOC-SPERMIDINE | | Synonyms: | 1,6-bis-Boc-1,6,10-triazadecane;N1,N5-bis-Boc-sperMidine;BIS- BOC-SPERMIDINE;N1,N5-Bis-Boc-sperMidine;N1,N5-Bis-Boc-spermidine >=95.0% (TLC);Carbamic acid, (3-aminopropyl)[4-[[(1,1-dimethylethoxy)carbonyl]amino]butyl]-, 1,1-dimethylethyl ester;Tert-Butyl (3-aminopropyl)(4-((tert-butoxycarbonyl)amino)butyl)carbamate;tert-butyl N-(3-aminopropyl)-N-(4-{[(tert-butoxy)carbonyl]amino}butyl)carbamate | | CAS: | 68076-39-1 | | MF: | C17H35N3O4 | | MW: | 345.48 | | EINECS: | | | Product Categories: | | | Mol File: | 68076-39-1.mol | 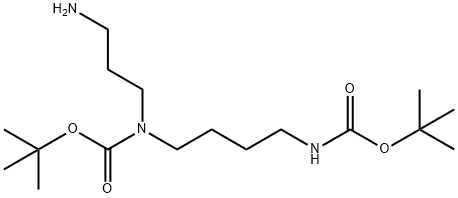 |
| | BIS- BOC-SPERMIDINE Chemical Properties |
| Boiling point | 462.0±38.0 °C(Predicted) | | density | 1.028±0.06 g/cm3(Predicted) | | storage temp. | -20°C | | pka | 12.87±0.46(Predicted) | | BRN | 2059741 |
| WGK Germany | 3 | | HS Code | 29225090 |
| | BIS- BOC-SPERMIDINE Usage And Synthesis |
| Description | N1,N5-Bis-Boc-spermidine is a linker containing an amino group with two Boc-protected amino groups. The amino group is reactive with carboxylic acids, activated NHS esters, carbonyls (ketone, aldehyde) etc. The Boc group can be deprotected under mild acidic conditions to form the free amine. | | Chemical Properties | Light yellow viscid liquid |
| | BIS- BOC-SPERMIDINE Preparation Products And Raw materials |
| Raw materials | 3-BOC-(4-BOC-AMINOBUTYL)AMINOPROPANENITRILE-->N(1)-benzyloxycarbonyl-N(8)-butoxycarbonylspermidine-->Carbamic acid, [3-(1,3-dihydro-1,3-dioxo-2H-isoindol-2-yl)propyl][4-[[(1,1-dimethylethoxy)carbonyl]amino]butyl]-, 1,1-dimethylethyl ester (9CI)-->Carbamic acid, [4-[(2-cyanoethyl)amino]butyl]-, 1,1-dimethylethyl ester-->tert-Butyl N-(4-aminobutyl)carbamate-->N-Boc-1,3-propanediamine-->tert-butyl (4-((3-chloropropyl)amino)-4-oxobutyl)carbamate(WXG01289)-->N-(2-Cyanoethyl)-1,4-diaminobutane-->BOC-GAMMA-ABU-OH-->Di-tert-butyl dicarbonate-->2-(tert-Butoxycarbonyloxyimino)-2-phenylacetonitrile |
|