|
|
| | CINNAMYLIDENEACETOPHENONE Basic information |
| Product Name: | CINNAMYLIDENEACETOPHENONE | | Synonyms: | 1,5-diphenyl-4-pentadien-1-one;2,4-Pentadien-1-one, 1,5-diphenyl-;5-phenyl-2,4-pentadienophenone;5-phenyl-4-pentadienophenone;alpha-Cinnamylideneacetophenone;cinnamalacetophenone;CINNAMYLIDENEACETOPHENONE;1,5-DIPHENYL-PENTA-2,4-DIEN-1-ONE | | CAS: | 614-57-3 | | MF: | C17H14O | | MW: | 234.29 | | EINECS: | 210-385-9 | | Product Categories: | Miscellaneous | | Mol File: | 614-57-3.mol | 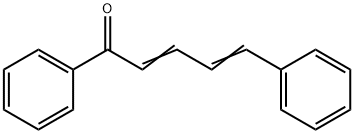 |
| | CINNAMYLIDENEACETOPHENONE Chemical Properties |
| Provider | Language |
|
ALFA
| English |
| | CINNAMYLIDENEACETOPHENONE Usage And Synthesis |
| Description | Colorless. viscous liquid. B.P. 239℃. Sp,Gr. 0.895. The aldehyde is conventionally sold in reduced strength, either 50% or 70% aldehyde content. The diluent may be the corresponding acetal, or an odorless solvent, e. g. Diethyl - phthalate. | | Uses | Cinnamylideneacetophenone is used in perfume compositions as a floralizing aldehydic topnote ingredient, primariIy in Muguet-Lily fragrances, but also in Rose, Peony (where the Geranyl-isomer is claimed to lx superior), and various modifications of modern-aldehydic fragrance types. Occasionally used in flavor compositions for floral or fruity types, and in Rose type flavors. The concentration is usually mere traces in the finished product. | | Synthesis | Cinnamylideneacetophenone is prepared from Citronellol, reacted with Sodium methylate or Sodium iso-propylate. The acetal is then prepared by reaction with Chlorodimethylacetal. The resulting acetal is finally hydrolyzed with diluted oxalic acid to yield the aldehyde. |
| | CINNAMYLIDENEACETOPHENONE Preparation Products And Raw materials |
|