17-PHENYL TRINOR PGF2ALPHA-IPR manufacturers
|
| | 17-PHENYL TRINOR PGF2ALPHA-IPR Basic information |
| Product Name: | 17-PHENYL TRINOR PGF2ALPHA-IPR | | Synonyms: | 17-phenyl-18,19,20-trinor-prostaglandinf2alpha-1-isopropylester;9ALPHA,11ALPHA,15S-TRIHYDROXY-17-PHENYL-18,19,20-TRINOR-PROSTA-5Z,13E-DIEN-1-OIC ACID, ISOPROPYL ESTER;17-PHENYL TRINOR PROSTAGLANDIN F2ALPHA ISOPROPYL ESTER;17-PHENYL TRINOR PGF2ALPHA-IPR;7-[(1R,2R,3R,5S)-3,5-Dihydroxy-2-[(1E,3S)-3-hydroxy-5-phenyl-1-penten-1-yl]cyclopentyl]-5-heptenoic acid 1-methylethyl ester;Bimatoprost isopropyl ester;17-phenyl trinor Prostaglandin F2α isopropyl ester;17-phenyl trinor PGF2α isopropyl ester | | CAS: | 130209-76-6 | | MF: | C26H38O5 | | MW: | 430.58 | | EINECS: | | | Product Categories: | | | Mol File: | 130209-76-6.mol | 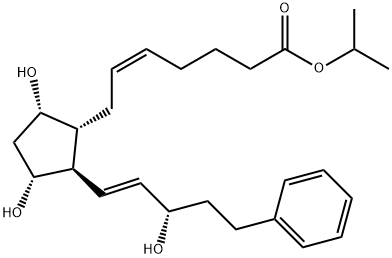 |
| | 17-PHENYL TRINOR PGF2ALPHA-IPR Chemical Properties |
| Boiling point | 573.4±50.0 °C(Predicted) | | density | 1.141 | | storage temp. | Store at -20°C | | solubility | DMF: 30 mg/ml; DMSO: 30 mg/ml; Ethanol: 50 mg/ml; Ethanol:PBS (pH 7.2) (1:1): 1 mg/ml | | pka | 14.25±0.20(Predicted) |
| | 17-PHENYL TRINOR PGF2ALPHA-IPR Usage And Synthesis |
| Description | 17-phenyl trinor PGF2α N-ethyl amide is an F-series prostaglandin analog which has been approved for use as an ocular hypotensive drug, sold under the Allergan trade name Bimatoprost. The N-ethyl amide prostaglandin prodrugs are converted to the active free acid more slowly than the analogous prostaglandin ester prodrugs such as latanoprost. This product is the isopropyl ester of the free acid prostaglandin which corresponds to Bimatoprost. The free acid, 17-phenyl trinor PGF2α, is a potent FP receptor agonist. In human and animal models of glaucoma, FP receptor agonist activity corresponds very closely with intraocular hypotensive activity. The 17-phenyl trinor PGF2α isopropyl ester derivative was examined for IOP-lowering activity during the development of latanoprost. At the dose of 3 μg/eye in the monkey, 17-phenyl trinor PGF2α isopropyl ester was the most potent analog tested in reducing IOP, lowering the IOP 1.3 mm Hg below the level achieved by latanoprost. However, this derivative was also significantly more irritating to the eye than latanoprost. | | Uses | PhDH 100A can be useful in the determination of prostaglandin analogs in cosmetic products by high performance liquid chromatography with tandem mass spectrometry. | | Definition | ChEBI: 17-phenyl-trinor-PGF2alpha isopropyl ester is a prostanoid. |
| | 17-PHENYL TRINOR PGF2ALPHA-IPR Preparation Products And Raw materials |
|