|
|
| | Propargyl-PEG5-CH2CO2tBu Basic information |
| Product Name: | Propargyl-PEG5-CH2CO2tBu | | Synonyms: | Propargyl-PEG5-CH2CO2tBu;Propargyl-PEG4-O-C1-Boc;3,6,9,12,15-Pentaoxaoctadec-17-ynoic acid, 1,1-dimethylethyl ester;Propargyl-PEG5-CH2COOtBu;Propargyl-PEG5-CH2COOH t-Bu Ester;Propargyl-PEG4-CH2COOtBu/3,6,9,12,15-Pentaoxaoctadec-17-ynoic acid, 1,1-dimethylethyl ester;tert-butyl 2-[2-[2-[2-(2-prop-2-ynoxyethoxy)ethoxy]ethoxy]ethoxy]acetate | | CAS: | 2098489-63-3 | | MF: | C17H30O7 | | MW: | 346.42 | | EINECS: | | | Product Categories: | | | Mol File: | 2098489-63-3.mol | 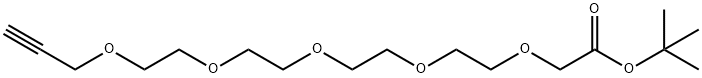 |
| | Propargyl-PEG5-CH2CO2tBu Chemical Properties |
| Boiling point | 406.8±40.0 °C(Predicted) | | density | 1.055±0.06 g/cm3(Predicted) | | form | Liquid | | color | Light yellow to yellow |
| | Propargyl-PEG5-CH2CO2tBu Usage And Synthesis |
| Description | Propargyl-PEG5-CH2CO2tBu enables the formation of triazole linkages with azide compounds or biomolecules in copper catalyzed Click Chemistry reactions. The t-butyl group can be hydrolyzed under acidic conditions. |
| | Propargyl-PEG5-CH2CO2tBu Preparation Products And Raw materials |
|