|
|
| | 16-EPIESTRIOL Basic information |
| Product Name: | 16-EPIESTRIOL | | Synonyms: | 16beta,17beta-Estriol;16-Epiestratriol;3,16beta,17beta-Estriol;5(10)-triene-3,16,17-triol,(16beta,17beta)-estra-3;Actriol;Epiestriol;Epioestriol;Estra-1,3,5(10)-triene-3,16,17-triol, (16beta,17beta)- | | CAS: | 547-81-9 | | MF: | C18H24O3 | | MW: | 288.38 | | EINECS: | 208-937-9 | | Product Categories: | Steroids;Intermediates & Fine Chemicals;Metabolites & Impurities;Pharmaceuticals | | Mol File: | 547-81-9.mol | 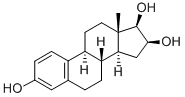 |
| | 16-EPIESTRIOL Chemical Properties |
| Melting point | 289-2910C | | Boiling point | 370.61°C (rough estimate) | | density | 1.1285 (rough estimate) | | refractive index | 1.5000 (estimate) | | storage temp. | -20°C Freezer | | solubility | DMSO (Slightly), Ethanol (Slightly, Sonicated), Methanol (Slightly, Heated) | | form | Solid | | pka | 10.25±0.70(Predicted) | | color | White to Off-White |
| | 16-EPIESTRIOL Usage And Synthesis |
| Chemical Properties | Crystalline Solid | | Uses | estrogen | | Uses | 16-Epiestriol is a metabolite of Estradiol. | | Definition | ChEBI: A 3-hydroxy steroid that is 17beta-estradiol substituted by a beta-hydroxy group at position 16. | | Biochem/physiol Actions | 16-Epiestriol is found naturally in various animals, plants, and fungal yeast. It potentially inhibits the growth of colistins and carbapenem-resistant A. baumannii |
| | 16-EPIESTRIOL Preparation Products And Raw materials |
|