| Company Name: |
BOC Sciences
|
| Tel: |
|
| Email: |
info@bocsci.com |
| Products Intro: |
Product Name:Muzolimine
CAS:55294-15-0
Purity:98% Remarks:Muzolimine is a pyrazole and high-ceiling loop diuretic with long duration and high capacity of action. It was used for kidney failure and hypertension but was withdrawn worldwide because of severe ne
|
|
| | Muzolimine Basic information |
| Product Name: | Muzolimine | | Synonyms: | 3-Amino-1-(3,4-dichloro-α-methylbenzyl)-2-pyrazolin-5-one;BAY-g-2821;Edrul;3-amino-1-(1-(3,4-dichlorophenyl)ethyl)-1H-pyrazol-5(4H)-one;Muzolimine;3H-Pyrazol-3-one, 5-amino-2-[1-(3,4-dichlorophenyl)ethyl]-2,4-dihydro-;Muzolimina;Muzoliminum | | CAS: | 55294-15-0 | | MF: | C11H11Cl2N3O | | MW: | 272.13 | | EINECS: | 2595732 | | Product Categories: | | | Mol File: | 55294-15-0.mol | 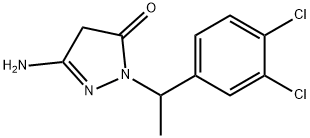 |
| | Muzolimine Chemical Properties |
| Melting point | 127-129° | | Boiling point | 401.4±55.0 °C(Predicted) | | density | 1.5374 (rough estimate) | | refractive index | 1.5500 (estimate) | | pka | 3.30±0.50(Predicted) | | form | Solid | | color | Light yellow to yellow |
| Toxicity | LD50 in mice, rats, dogs, rabbits (mg/kg): 1794, 1559, 2000, 1250 orally (Lorke, Mürmann) |
| | Muzolimine Usage And Synthesis |
| Description | Muzolimine is a structurally novel, pyrazolone diuretic with a high-ceiling profile.
It is somewhat slower in onset than furosemide, but has a more prolonged effect,
similar to the thiazides. Muzolimine has been shown to be effective in edema of
cardiac, hepatic and renal origin. It also appears to be effective as an antihypertensive
agent. | | Originator | Bayer (W. Germany) | | Uses | Diuretic; antihypertensive. | | Definition | ChEBI: Muzolimine is a dichlorobenzene. | | Manufacturing Process | 41 g of α-methyl-3,4-dichlorobenzylhydrazine, dissolved in absolute ethanol,
were added dropwise to a solution of 31.8 g of β-amino-β-ethoxyacrylic acid
ethyl ester and 1.5 g of p-toluenesulfonic acid in 150 ml of ethanol at room
temperature under nitrogen gas. After stirring for 2 hours and standing
overnight, the reaction solution was concentrated as far as possible on a
rotary evaporator. The residue which remained was dissolved in 2 N sodium
hydroxide solution. Any unconverted starting products or by-products were
extracted with ether. The aqueous phase was then brought to pH 5 with acetic
acid. The oil thereby produced was taken up in methylene chloride and the
organic phase was dried over Na2SO4. After evaporating off the solvent, the
reaction product crystallized out. It was recrystallized from methanol; melting
point 127°C to 129°C; yield 21 g (38.5% of theory). | | Brand name | EDRUL | | Therapeutic Function | Diuretic | | World Health Organization (WHO) | Reports of neurological adverse effects, including paraesthesiae
and paralyses, associated with prolonged use of high dosages of muzolimine, were
received shortly after its introduction in 1984. |
| | Muzolimine Preparation Products And Raw materials |
|