S-ADENOSYL-L-METHIONINE IODIDE SALT manufacturers
|
| | S-ADENOSYL-L-METHIONINE IODIDE SALT Basic information |
| Product Name: | S-ADENOSYL-L-METHIONINE IODIDE SALT | | Synonyms: | SAM-E IODIDE SALT;S-(5'-ADENOSYL)-L-METHIONINE IODIDE;S-ADENOSYL-L-METHIONINE IODIDE SALT;S-ADENOSYLMETHIONINE IODIDE;S-ADENOSYL METHIONINE IODIDE SALT;ADENOSYL-L-METHIONINE, S-IODIDE SALT;S-(5'-deoxy-5'-adenosyl)methionine iodide;S-adenosylmethione | | CAS: | 3493-13-8 | | MF: | C15H23IN6O5S | | MW: | 526.35 | | EINECS: | 222-486-5 | | Product Categories: | Nucleic acids | | Mol File: | 3493-13-8.mol | 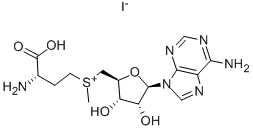 |
| | S-ADENOSYL-L-METHIONINE IODIDE SALT Chemical Properties |
| storage temp. | -20°C | | solubility | H2O: 100 mg/mL | | BRN | 4120787 |
| | S-ADENOSYL-L-METHIONINE IODIDE SALT Usage And Synthesis |
| Uses | S-(5′-Adenosyl)-L-methionine (SAM, AdoMet) is used as a primary methyl donor molecule in mammalian cell culture and the first step metabolite in methionine biosynthesis. | | Biochem/physiol Actions | Methyl donor; cofactor for enzyme-catalyzed methylations, including catechol O-methyltransferase (COMT) and DNA methyltransferases (DNMT). Although present in all cells, it is concentrated in liver where 85% of all methylation reactions occur. It is also involved in regulating liver function, growth, and response to injury. |
| | S-ADENOSYL-L-METHIONINE IODIDE SALT Preparation Products And Raw materials |
|