| Company Name: |
TargetMol Chemicals Inc.
|
| Tel: |
4008200310 |
| Email: |
marketing@tsbiochem.com |
| Products Intro: |
Product Name:Butopamine;Butopamine
CAS:66734-12-1
Purity:98% Package:5 mg
|
|
| | Butopamine Basic information |
| Product Name: | Butopamine | | Synonyms: | (R)-4-Hydroxy-α-[[(R)-3-(4-hydroxyphenyl)-1-methylpropylamino]methyl]benzyl alcohol;4-[(R)-3-[[(R)-2-(4-Hydroxyphenyl)-2-hydroxyethyl]amino]butyl]phenol;Compound LY-131126;LY-131126;Butopamine;Benzenemethanol, 4-hydroxy-α-[[[(1R)-3-(4-hydroxyphenyl)-1-methylpropyl]amino]methyl]-, (αR)- | | CAS: | 66734-12-1 | | MF: | C18H23NO3 | | MW: | 301.38 | | EINECS: | | | Product Categories: | | | Mol File: | 66734-12-1.mol | 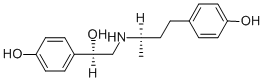 |
| | Butopamine Chemical Properties |
| Boiling point | 520.5±50.0 °C(Predicted) | | density | 1.189±0.06 g/cm3(Predicted) | | pka | 9.97±0.26(Predicted) |
| | Butopamine Usage And Synthesis |
| Originator | Butopamine,ZYF Pharm Chemical | | Definition | ChEBI: A 4-(1-hydroxy-2-{[4-(4-hydroxyphenyl)butan-2-yl]amino}ethyl)phenol in which both stereocentres have R configuration. It is the most active of the four diastereoisomers that make up the animal feed additive ractopamine. | | Manufacturing Process | A solution of 32.8 g (0.2 m) of methyl 2-(4-hydroxyphenyl)ethyl ketone and
42.6 g (0.2 m) of 1-(4-hydroxyphenyl)-2-aminoethanol in 380 ml of ethanol containing 3.8 g of 5% palladium on carbon was stirred for 16 h at 60°C
under hydrogen at 55 psi. The reaction mixture was then filtered, and the
filtrate was diluted by addition of 350 ml of water. The aqueous mixture was
concentrated to a volume of about 400 ml, and then washed with
dichloromethane.
The aqueous mixture was acidified by addition of 50 ml of conc. hydrochloric
acid. After standing at room temperature for 2 h, the aqueous acid mixture
was filtered and the filter cake was washed with 40 ml of ice water and dried
at 50°C in vacuum to provide 47.0 g of 1-(4-hydroxyphenyl-2-
propylamino]ethanol hydrochloride, melting point 124-129°C.
The 1-(4-hydroxyphenyl)-2-[1-methyl-3-(4-hydroxyphenyl)propylamino]
ethanol was obtained by treatment of 1-(4-hydroxyphenyl-2-
propylamino]ethanol hydrochloride with sodium hydroxide. | | Therapeutic Function | Cardiotonic |
| | Butopamine Preparation Products And Raw materials |
|