5-HYDROXYTRYPTOPHOL manufacturers
- 5-Hydroxytryptophol
-
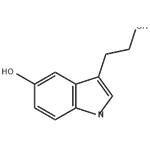
- $0.00 / 1KG
-
2024-07-16
- CAS:154-02-9
- Min. Order: 1KG
- Purity: 99.0%
- Supply Ability: 1000KG
- 5-HYDROXYTRYPTOPHOL
-
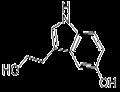
- $0.01 / 1KG
-
2019-12-25
- CAS:154-02-9
- Min. Order: 1KG
- Purity: 95%-99%
- Supply Ability: 1kg; 100kg; 500kg
|
| | 5-HYDROXYTRYPTOPHOL Basic information |
| | 5-HYDROXYTRYPTOPHOL Chemical Properties |
| Melting point | 111-113° | | Boiling point | 435.2±30.0 °C(Predicted) | | density | 1.356±0.06 g/cm3(Predicted) | | storage temp. | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C | | solubility | Chloroform (Slightly), Methanol (Slightly) | | pka | 10.07±0.40(Predicted) | | form | Solid | | color | White to Light Red |
| RTECS | NL8512500 | | HS Code | 2933998090 |
| | 5-HYDROXYTRYPTOPHOL Usage And Synthesis |
| Chemical Properties | Off-White Crystalline Powder | | Uses | A metabolite of Tryptophan (T947200). | | Definition | ChEBI: 5-Hydroxytryptophol is a member of indoles. | | Hazard | A reproductive hazard. | | Biological Activity | 5-hydroxy tryptophol is a metabolite of tryptophan.tryptophan (encoded by the codon ugg) is an α-amino acid that is used in the biosynthesis of proteins. tryptophan has an α-amino group, an α-carboxylic acid group, and a side chain indole, demonstrating it as a non-polar, aromatic amino acid. tryptophan is critical in humans, but the body cannot synthesize it and therefore tryptophan must be obtained from the diet. tryptophan is also found to be a precursor to the neurotransmitters serotonin and melatonin. | | in vitro | in previous studies, 5-hydroxy tryptophol was identified as a metabolite of tryptophan that was formed by the alcohol dehydrogenase-catalyzed reduction of the serotonin intermediate, 5-hydroxyindoleacetaldehyde. moreover, depending on the tissue nad/nadh ratio, the acetaldehyde intermediate could also be oxidized by aldehyde dehydrogenase to form 5-hydroxyindoleacetic acid. therefore, the ratio of 5-hydroxy tryptophol to 5-hydroxyindoleacetic acid could be used as a biomarker for recent alcohol consumption [1,2]. | | references | [1] yokoyama, m. t. and carlson, j.r. dissimilation of tryptophan and related indolic compounds by ruminal microorganisms in vitro. appl.microbiol. 27(3), 540-548 (1974).
[2] beck, o. ,stephanson, n.,btthcer, m., et al. biomarkers to disclose recent intake of alcohol: potential of 5-hydroxytryptophol glucuronide testing using new direct uplc-tandem ms and elisa methods. alcohol & alcoholism 42(4), 321-325 (2007). |
| | 5-HYDROXYTRYPTOPHOL Preparation Products And Raw materials |
|