|
|
| | 2-(2-aminobut-3-enyl)propanedioic acid Basic information |
| Product Name: | 2-(2-aminobut-3-enyl)propanedioic acid | | Synonyms: | 2-[(2RS)-2-Aminobut-3-enyl]propanedioic acid;Vigabatrin impurity E;2-(2-Aminobut-3-enyl)malonic Acid;Vigabatrin Impurity 5(Vigabatrin EP Impurity E);2-(2-aminobut-3-enyl)propanedioic acid;Vigabatrin EP Impurity E;Propanedioic acid, 2-(2-amino-3-buten-1-yl)-;2-(2-Aminobut-3-en-1-yl)malonic acid | | CAS: | 1378466-25-1 | | MF: | C7H11NO4 | | MW: | 173.17 | | EINECS: | | | Product Categories: | | | Mol File: | 1378466-25-1.mol | 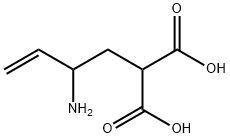 |
| | 2-(2-aminobut-3-enyl)propanedioic acid Chemical Properties |
| Boiling point | 420.1±45.0 °C(Predicted) | | density | 1.303±0.06 g/cm3(Predicted) | | pka | 2.58±0.34(Predicted) | | InChI | InChI=1S/C7H11NO4/c1-2-4(8)3-5(6(9)10)7(11)12/h2,4-5H,1,3,8H2,(H,9,10)(H,11,12) | | InChIKey | AIFHTSLGFACKRW-UHFFFAOYSA-N | | SMILES | C(O)(=O)C(CC(N)C=C)C(O)=O |
| | 2-(2-aminobut-3-enyl)propanedioic acid Usage And Synthesis |
| Description | 2-(2-aminobut-3-enyl)propanedioic acid is a naturally occurring non-protein amino acid, also known as l-allyl glycine. This acid is involved in various metabolic processes and is found in many plants, animals, and microorganisms. In 1931, Heilbron and Hodge first identified the L-allylglycine component in plant extracts. At the time, researchers noted that L-allyl glycine is a very potent inhibitor of alanine aminotransferase, and later found that it also inhibits cystathionine γ-synthetase. | | Uses | 2-(2-Aminobut-3-enyl)malonic Hydrochloric Acid, is a derivative of Vigabatrin (V253000), which is an antiepileptic drug that inhibits the catabolism of gamma-aminobutyric acid (GABA) by irreversibly inhibiting GABA transaminase. |
| | 2-(2-aminobut-3-enyl)propanedioic acid Preparation Products And Raw materials |
|