|
|
| | 4,5-Dichloro-1,2,3-dithiazolium chloride Basic information |
| Product Name: | 4,5-Dichloro-1,2,3-dithiazolium chloride | | Synonyms: | 4,5-Dichloro-1,2,3-dithiazol-1-ium chloride;Appel salt;4,5-Dichloro-1,2,3-dithiazolium chloride;D14145;4,5-DICHLORO-1,2,3-DITHIAZOLIUM;4,5-Dichloro-1,2,3-dithiazol-2-ylium chloride;4,5-Dichlorodithiazol-2-ium chloride;Appel's Salt | | CAS: | 75318-43-3 | | MF: | C2Cl3NS2 | | MW: | 208.5 | | EINECS: | | | Product Categories: | | | Mol File: | 75318-43-3.mol | 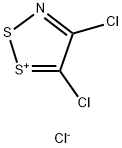 |
| | 4,5-Dichloro-1,2,3-dithiazolium chloride Chemical Properties |
| Melting point | 172° (dec) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | form | solid | | color | Brown |
| | 4,5-Dichloro-1,2,3-dithiazolium chloride Usage And Synthesis |
| Description | 4,5-Dichloro-1,2,3-dithiazolium chloride is an useful organosulfur compound. It is the chloride salt of the 4,5-dichloro-1,2,3-dithiazolium cation. It is a green solid that is poorly soluble in organic solvents. Most primary arylamines react readily with 4,5-dichloro-1,2,3-dithiazolium chloride to give, after treatment with tertiary amine base (2 equiv.), the corresponding N-aryl-4-chloro-5H-1,2,3-dithiazolimines in good to excellent yields. This compound is commonly used to synthesize monocyclic 1,2,3-dithiazoles which act as fungicides, antibacterials, antivirals or anticancer agents[1-2].
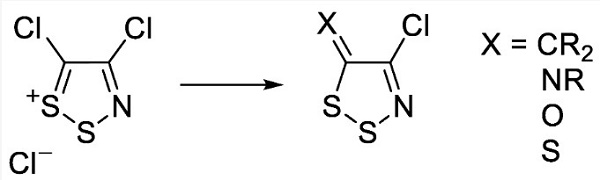 | | Uses | Synthon in heterocyclic chemistry; in conversion of alcohols and carboxylic acids to esters. | | References | [1] Kalogirou A, et al. The Reaction of 4,5-Dichloro-1,2,3-dithiazolium Chloride with Sulfimides: A New Synthesis of N-Aryl-1,2,3-dithiazolimines. Molecules, 2009; 14: 2356–2362.
[2] Plakas K, et al. Reaction of 4,5-Dichloro-1,2,3-dithiazolium Chloride with 2-(Phenylsulfonyl)acetonitrile. Molbank, 2022; 2022: M1322. |
| | 4,5-Dichloro-1,2,3-dithiazolium chloride Preparation Products And Raw materials |
|