- Pentapeptide-18
-

- $30.00 / 1kg
-
2024-09-30
- CAS:64963-01-5
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 2000kg
|
| | (D-Ala2)-Leu-Enkephalin Basic information |
| Product Name: | (D-Ala2)-Leu-Enkephalin | | Synonyms: | H-TYR-D-ALA-GLY-PHE-LEU-OH;D-ALA2-LEU5-ENKEPHALIN;[D-ALA2]-LEUCINE ENKEPHALIN;TYR-D-ALA-GLY-PHE-LEU;N-[N-[N-(N-L-tyrosyl-D-alanyl)glycyl]-L-phenylalanyl]-L-leucine;(D-alanine2)leucine enkephalin;L-Tyr-D-Ala-Gly-L-Phe-L-Leu-OH;Tyr-D-Ala-Gly-Phe-Leu-OH | | CAS: | 64963-01-5 | | MF: | C29H39N5O7 | | MW: | 569.65 | | EINECS: | 265-291-0 | | Product Categories: | | | Mol File: | 64963-01-5.mol | 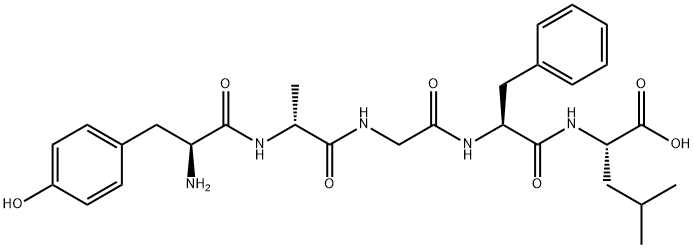 |
| | (D-Ala2)-Leu-Enkephalin Chemical Properties |
| Melting point | 160-163.5 °C | | Boiling point | 991.9±65.0 °C(Predicted) | | density | 1.257±0.06 g/cm3(Predicted) | | storage temp. | Keep in dark place,Sealed in dry,Store in freezer, under -20°C | | solubility | DMF (Slightly), DMSO (Slightly) | | form | Solid | | pka | 3.40±0.10(Predicted) | | color | White to Off-White | | InChIKey | ZHUJMSMQIPIPTF-JMBSJVKXSA-N | | SMILES | C(O)(=O)[C@H](CC(C)C)NC(=O)[C@H](CC1=CC=CC=C1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@H](CC1=CC=C(O)C=C1)N |
| | (D-Ala2)-Leu-Enkephalin Usage And Synthesis |
| Definition |
The leucine-enkephalin (Tyr-Gly-Gly-Phe-Leu, Leu-enk) is a natural endogenous ligand for opiate receptors. Leu-enk is rapidly metabolized in vivo and in vitro[1]. (D-Ala2)-Leu-Enkephalin, a delta opioid agonist, is a degradation resistant long-acting Leu-enkephalin.
| | benefits |
(D-Ala2)-Leu-Enkephalin is widely distributed in the human body and has essential physiological functions. It has a unique effect on brain cells and can activate sleeping brain cells and have a good recovery effect on the sequelae caused by brain damage. It plays an important role in immune function, including increasing the activity of the immune system, T lymphocytes, NK cells, macrophages, antiviral, and the release of gamma interferon. It can participate in immune regulation by promoting the secretion of endocrine cells by activated immune cells, thereby affecting DNA's gene regulation and expression functions.
| | References |
[1] H. Hasegawa, S. Baba, Y. Shinohara. “Synthesis of [D-Ala2]Leu-enkephalin and [D-Ala2, D-Leu5]Leu-enkephalin with high specific tritiated activity in the leucine residue.” Journal of The Chemical Society-perkin Transactions 1 12 1 (1990): 2641–2644.
|
| | (D-Ala2)-Leu-Enkephalin Preparation Products And Raw materials |
|