|
|
| | Ethyl 3-(2,5-difluorophenyl)-3-oxopropanoate Basic information |
| Product Name: | Ethyl 3-(2,5-difluorophenyl)-3-oxopropanoate | | Synonyms: | ETHYL 3-(2,5-DIFLUOROPHENYL)-3-OXOPROPANOATE;ETHYL 2,5-DIFLUOROBENZOYLACETATE;3-(2,5-Difluoro-phenyl)-3-oxo-propionic acid ethyl ester;Ethyl 3-(2,5-difluorophenyl);2-fluoro-3-(3-fluorophenyl)-3-oxopropanoic acid ethyl ester;Ethyl 3-(2,5-difluorophenyl)-3-oxopropanoate 95+%;Benzenepropanoic acid, 2,5-difluoro-β-oxo-, ethyl ester;Ethyl 2,5-difluoro-β-oxobenzenepropanoate | | CAS: | 887267-53-0 | | MF: | C11H10F2O3 | | MW: | 228.19 | | EINECS: | | | Product Categories: | | | Mol File: | 887267-53-0.mol | 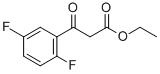 |
| | Ethyl 3-(2,5-difluorophenyl)-3-oxopropanoate Chemical Properties |
| Boiling point | 275.7±25.0 °C(Predicted) | | density | 1.251±0.06 g/cm3(Predicted) | | pka | 10.28±0.50(Predicted) |
| | Ethyl 3-(2,5-difluorophenyl)-3-oxopropanoate Usage And Synthesis |
| Uses | Ethyl 3-(2,5-difluorophenyl)-3-oxopropanoate is commonly utilized in organic synthesis as C-nucleophiles. One specific example is its reaction with the N-terminal electrophile CF3CHN2, resulting in the formation of CF3-heterocycles, namely ethyl 6-fluoro-4-oxo-1-(2,2,2-trifluoroethyl)-1,4-dihydrocinnoline-3-carboxylate[1].
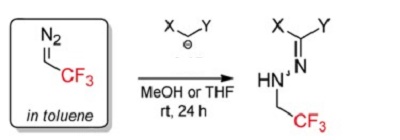 | | References | [1] Arkhipov A, et al. Unexpected Reactivity of Trifluoromethyl Diazomethane (CF3CHN2):
Electrophilicity of the Terminal N?Atom. Organic Letters, 2016; 18: 3406–3409. |
| | Ethyl 3-(2,5-difluorophenyl)-3-oxopropanoate Preparation Products And Raw materials |
|