- Hexanoic Anhydride
-

- $5.00 / 1KG
-
2024-10-11
- CAS:2051-49-2
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 10000kg
- Caproic anhydride
-
- $390.00 / 1Kg
-
2024-10-06
- CAS:2051-49-2
- Min. Order: 0.100Kg
- Purity: 99
- Supply Ability: 1000 Kg
- HEXANOIC ANHYDRIDE
-

- $0.00 / 25KG
-
2023-11-13
- CAS:2051-49-2
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 50000KG/month
|
| | HEXANOIC ANHYDRIDE Basic information |
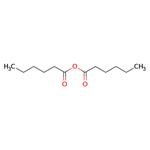
| Product Name: | HEXANOIC ANHYDRIDE | | Synonyms: | N-BUTYLACETIC ANHYDRIDE;N-CAPRONIC ANHYDRIDE;N-CAPROIC ANHYDRIDE;N-HEXANOIC ANHYDRIDE;N-HEXOIC ANHYDRIDE;PENTYLFORMIC ANHYDRIDE;N-CAPROICANHYDRIDE=HEXANOICANHYDRIDE;Bis(hexanoic)anhydride | | CAS: | 2051-49-2 | | MF: | C12H22O3 | | MW: | 214.3 | | EINECS: | 218-121-4 | | Product Categories: | Pharmaceutical Intermediates | | Mol File: | 2051-49-2.mol |  |
| | HEXANOIC ANHYDRIDE Chemical Properties |
| Melting point | -40 °C | | Boiling point | 246-248 °C (lit.) | | density | 0.928 g/mL at 20 °C (lit.) | | vapor pressure | 2.9Pa at 25℃ | | refractive index | n20/D 1.428(lit.) | | Fp | >230 °F | | storage temp. | Store below +30°C. | | solubility | ethanol: soluble1g/10 mL, clear, colorless | | form | Liquid | | color | Clear colorless to light yellow | | explosive limit | 0.7%(V) | | Water Solubility | Hydrolyzes in water. | | Sensitive | Moisture Sensitive | | BRN | 1776561 | | LogP | 4.45 at 25℃ | | CAS DataBase Reference | 2051-49-2(CAS DataBase Reference) | | EPA Substance Registry System | Hexanoic acid, anhydride (2051-49-2) |
| Hazard Codes | C | | Risk Statements | 34 | | Safety Statements | 26-36/37/39-45 | | RIDADR | UN 3265 8/PG 2 | | WGK Germany | 3 | | TSCA | Yes | | HazardClass | 8 | | PackingGroup | III | | HS Code | 29159000 |
| | HEXANOIC ANHYDRIDE Usage And Synthesis |
| Chemical Properties | clear colorless to light yellow liquid | | Uses | Hexanoic anhydride has been used in:
- green synthesis of esters of acyclovir (acyclovir prodrugs)
- preparation of hexanoyl-modified chitosan nanoparticles and chitosan-based polymeric surfactants via N-acylation of chitosans
| | Uses | Hexanoic anhydride was used in:
- green synthesis of esters of acyclovir (acyclovir prodrugs)
- preparation of hexanoyl-modified chitosan nanoparticles
- preparation of chitosan-based polymeric surfactants via N-acylation of chitosans
| | Uses | Hexanoic Anhydride, is used as a reactant in the total synthesis of acremomannolipin A via steroselective β-mannosylation of 4,6,-O-benzylidene-protected mannosyl sulfoxide with a D-mannitol derivative. | | Preparation | To an ice-cooled flask containing 116 gm (1.0 mole) of n-caproic acid is added 21.0-23.10 gm (0.5-0.55 mole) of ketene at a rate of 0.45 mole/hr. The reaction mixture is fractionally distilled at atmospheric pressure to afford a forecut of acetone, acetic acid, and acetic anhydride. The oil bath is raised to 220°C over a 1-hr period, kept there for 3 hr to ensure complete removal of acetic acid, and then cooled. The distillation is continued under reduced pressure to afford 86-95 gm (80-87%), b.p. 109- 112°C (3 mm Hg) and b.p. 118-121°C (6 mm Hg).
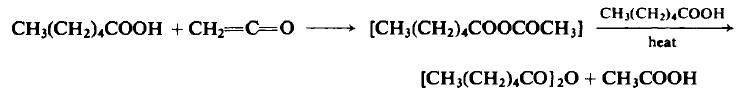 | | Flammability and Explosibility | Not classified |
| | HEXANOIC ANHYDRIDE Preparation Products And Raw materials |
|