|
|
| | Fendosal Basic information |
| Product Name: | Fendosal | | Synonyms: | hp129;5-(4,5-dihydro-2-phenyl-3h-benz[e]indol-3-yl)salicylic acid;Fendozal;P 71-0129;2-hydroxy-5-{2-phenyl-3H,4H,5H-benzo[e]indol-3-yl}benzoic acid;2-Hydroxy-5-(2-phenyl-4,5-dihydro-3H-benzo[e]indol-3-yl)benzoic acid;FENDOSAL;3-(3-carboxy-4-hydroxyphenyl)-2-phenyl-4,5-dihydro-3h-benz(e)indole | | CAS: | 53597-27-6 | | MF: | C25H19NO3 | | MW: | 381.42 | | EINECS: | | | Product Categories: | | | Mol File: | 53597-27-6.mol | 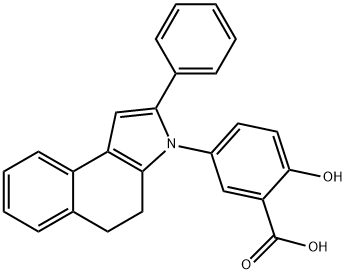 |
| | Fendosal Chemical Properties |
| Melting point | 223-225° (dec) | | Boiling point | 508.82°C (rough estimate) | | density | 1.2182 (rough estimate) | | refractive index | 1.6000 (estimate) | | pka | 2.40±0.10(Predicted) | | CAS DataBase Reference | 53597-27-6(CAS DataBase Reference) |
| Toxicity | LD50 in mice, rats, rabbits (mg/kg): 740, 450, 560 orally; in mice (mg/kg): 510 i.p. (Lassman) |
| | Fendosal Usage And Synthesis |
| Originator | Alnovin,Hoechst | | Uses | Anti-inflammatory. | | Definition | ChEBI: Fendosal is a benzoindole that is 4,5-dihydro-3H-benzo[e]indole in which the nitrogen is substituted by a 3-carboxy-4-hydroxyphenyl group. A non-narcotic analgesic and non-steroidal anti-inflammatory drug, it has greater analgesic and inflammatory responses than aspirin but with less gastrointestinal toxicity. It has a role as a non-steroidal anti-inflammatory drug and a non-narcotic analgesic. It is a member of pyrroles, a benzoindole and a monohydroxybenzoic acid. | | Manufacturing Process | To a stirred refluxing solution of 20.2 g (0.1 mole) of 1-(1-pyrrolidino)-3,4-
dihydronaphthalene and 50 ml of toluene was added dropwise during 30 minutes under nitrogen a solution of 20.1 g (0.1 mole) of phenacyl bromide in
65 ml of dry toluene. The mixture was heated under reflux for 6 hours, diluted
with 50 ml of water, refluxed for 4 hours, and cooled. The layers were
separated and the aqueous phase was extracted with benzene. The organic
solution was dried over sodium sulfate and concentrated to a semi-solid.
Trituration with cold 30°-60° petroleum ether gave 23.6 g (78%) of solid, 2-
phenacyl-1-tetralone, MP: 73°-76°C. Recrystallization from 60°-90° petroleum
ether raised the melting point to 87°-88°C.
A mixture of 20.0 g (0.076 mole) of 2-phenacyl-1-tetralone, 11.6 g (0.076
mole) of 5-aminosalicyclic acid, and 70 ml of glacial acetic acid was heated
under reflux for 4 hours, cooled, diluted with 10 ml of water and filtered. The
filter cake was washed with water and dried to provide 15.5 g of solid, 5-(4,5-
dihydro-2-phenyl-3H-benz[e]indol-3-yl)-2-hydroxy-benzoic acid. MP: 215°-
218°C. Recristallization from benzene-cyclohexane gave 6.5 g (22%) of yellow
crystals, MP: 245°-247°C. | | Brand name | Alnovin(Hoechst-Roussel). | | Therapeutic Function | Analgesic, Antiinflammatory, Antipyretic |
| | Fendosal Preparation Products And Raw materials |
|