|
|
| | narciclasine Basic information |
| Product Name: | narciclasine | | Synonyms: | narciclasine;(13β)-1,19-Didehydro-2α,3β,4β,7-tetrahydroxy-11,12-dinor-5,19-secocrinan-6-one;(2S)-3,4,4aβ,5-Tetrahydro-2α,3β,4β,7-tetrahydroxy[1,3]dioxolo[4,5-j]phenanthridin-6(2H)-one;Lycorcidinol;(2S,3R,4S,4aR)-3,4,4a,5-Tetrahydro-2,3,4,7-tetrahydroxy-(1,3)dioxolo(4,5-j)phenanthridin-6(2H)-one;(2S-(2-alpha,3-beta,4-beta,4a-beta))-3,4,4a,5-tetrahydro-2,3,4,7-tetrahydroxy-(1,3)Dioxolo(4,5-j)phenanthridin-6(2H)-one;3,4,4a,5-Tetrahydro-2,3,4,7-tetrahydroxy-(1,3)dioxolo(4,5-j)phenanthridin-6(2H)-one;BRN 1087400 | | CAS: | 29477-83-6 | | MF: | C14H13NO7 | | MW: | 307.26 | | EINECS: | | | Product Categories: | | | Mol File: | 29477-83-6.mol | 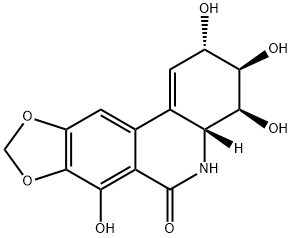 |
| | narciclasine Chemical Properties |
| Melting point | 250-252℃ | | Boiling point | 447.77°C (rough estimate) | | density | 1.85±0.1 g/cm3 (20 ºC 760 Torr) | | refractive index | 1.5300 (estimate) | | storage temp. | Store at -20°C | | solubility | DMSO: ≥10mg/mL | | pka | 7.72±0.70(Predicted) | | form | Solid | | color | white to off-white |
| Hazard Codes | Xn | | Risk Statements | 46 | | Safety Statements | 36 |
| | narciclasine Usage And Synthesis |
| Description | Narciclasine is a plant growth inhibitor that can be isolated from Narcissus bulbs. At 1 μM, it has been shown to induce apoptosis-mediated cytotoxicity in human cancer cells in vitro but not in normal fibroblasts. Narciclasine has been shown to regulate the Rho/Rho kinase/LIM kinase/cofilin signaling pathway by increasing GTPase RhoA activity, as well as inducing actin stress fiber formation in a RhoA-dependent manner. | | Description | The bulbs of several Narcissus species contain this alkaloid which crystallizes in
the form of pale yellow needles with a pronounced yellow-green fluorescence. It
is best purified by recrystallization from either acetic acid or an aqueous mixture
of methoxyethyl alcohol. It is dextrorotatory with [α]589 + 145° or [Qh64 +
983° (c 1.5, EtOH). The ultraviolet spectrum in neutral solution (EtOH) has
absorption maxima at 252, 302 and 329 mil, while that in alkaline solution
(0.01 N/NaOH) has the maxima at 219, 249,310 and 355 mil. The O-methyl
ether forms shiny needles which also exhibit a strong blue fluorescence. The tri_x0002_acetate has been prepared as an amorphous, non-crystallizable substance. With
FeC13 the alkaloid gives a violet colour. | | Uses | Narciclasine is an antiproliferative and pro-apoptotic inducer. | | Uses | Narciclasine is reasonably abundant in some Narcissus spp. and has served as a very useful intermediate for synthetic conversion into (+)-pancratistatin and to conduct a series of structure–activity relationship studies . Bicolorine, another member of the narciclasine series, is an unusual, completely aromatized quaternary alkaloid with an N-methyl group. | | Definition | ChEBI: A natural product found in Narcissus pseudonarcissus. | | target | Immunology & Inflammation related | | storage | Store at -20°C | | References | Ceriotti., Nature, 213,595 (1967)
Piozzi et al., Tetrahedron, 24, 1119 (1968)
Revised structure:
Mondon, Krohn., Tetrahedron Lett., 2123 (1970)
Savona, Piozzi, Marino., Chern. Cornrnun., 1006 (1970)
Absolute configuration:
Fuganti, Mazza.,J. Chern. Soc., Chern. Cornrnun., 239 (1972)
Crystal structure:
Immirzi, Fuganti., J. Chern. Soc., Chern. Cornrnun., 240 (1972)
Stereochemistry:
Mondon, Krohn., Tetrahedron Lett., 2085 (1972) |
| | narciclasine Preparation Products And Raw materials |
|