(5R)-4-乙酰基-1-(4-氯-2-氟苯基)-5-环己基-1,5-二氢-3-羟基-2H-吡咯-2-酮
| 中文名称 | (5R)-4-乙酰基-1-(4-氯-2-氟苯基)-5-环己基-1,5-二氢-3-羟基-2H-吡咯-2-酮 |
|---|---|
| 中文同义词 | (5R)-4-乙酰基-1-(4-氯-2-氟苯基)-5-环己基-1,5-二氢-3-羟基-2H-吡咯-2-酮;(R)-4-乙酰基-1-(4-氯-2-氟苯基)-5-环己基-3-羟基-1H-吡咯-2(5H)-酮;化合物CCR2-RA-[R;(R)-4-乙酰基-1-(4-氯-2-氟苯基)-5-环己基-3-羟基-1,5-二氢-2H-吡咯-2-酮 |
| 英文名称 | 2H-Pyrrol-2-one, 4-acetyl-1-(4-chloro-2-fluorophenyl)-5-cyclohexyl-1,5-dihydro-3-hydroxy-, (5R)- |
| 英文同义词 | 2H-Pyrrol-2-one, 4-acetyl-1-(4-chloro-2-fluorophenyl)-5-cyclohexyl-1,5-dihydro-3-hydroxy-, (5R)-;(R)-4-acetyl-1-(4-chloro-2-fluorophenyl)-5-cyclohexyl-3-hydroxy-1,5-dihydro-2H-pyrrol-2-one;187305;(R)-4-Acetyl-1-(4-chloro-2-fluorophenyl)-5-cyclohexyl-3-hydroxy-1H-pyrrol-2(5H)-one;(5R)-4-Acetyl-1-(4-chloro-2-fluorophenyl)-5-cyclohexyl-1,5-dihydro-3-hydroxy-2H-pyrrol-2-one;CCR,Inhibitor,inhibit,CCR-2-RA-[R],CC chemokine receptor,CCR2RA[R],CCR2 RA [R;4-ACETYL-1-(4-CHLORO-2-FLUOROPHENYL)-5-CYCLOHEXYL-3-HYDROXY-5H-PYRROL-2-ONE |
| CAS号 | 512177-83-2 |
| 分子式 | C18H19ClFNO3 |
| 分子量 | 351.8 |
| EINECS号 | |
| 相关类别 | |
| Mol文件 | 512177-83-2.mol |
| 结构式 | 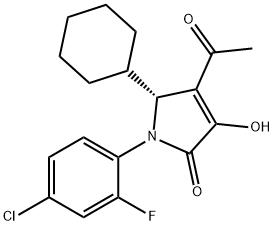 |
(5R)-4-乙酰基-1-(4-氯-2-氟苯基)-5-环己基-1,5-二氢-3-羟基-2H-吡咯-2-酮 性质
| 沸点 | 482.6±45.0 °C(Predicted) |
|---|---|
| 密度 | 1.383±0.06 g/cm3(Predicted) |
| 储存条件 | Store at -20°C |
| 溶解度 | DMF:30mg/mL; DMSO:30mg/mL; DMSO:PBS (pH 7.2) (1:3):0.25 mg/ml;乙醇:1mg/mL |
| 形态 | 结晶固体 |
| 酸度系数(pKa) | 4.50±1.00(Predicted) |
| 颜色 | 白色至米白色 |
|
CCR2 103 nM (IC 50 , in U2OS-CCR2 cells) |
The chemokine receptor CCR2 is a G protein-coupled receptor that is involved in many diseases characterized by chronic inflammation, and therefore a large variety of CCR2 small molecule antagonists has been developed. CCR2-RA-[R] displaces [ 125 I]CCL2 from CCR2 with an pIC 50 value of 6.1. The pK D of CCR2-RA-[R] for CCR2 and CCR5 is 8.8±0.1 and 7.0±0.1, respectively. CCR2-RA-[R] inhibits CCR2 non-competitively by blocking activation-associated conformational changes and formation of the G protein-binding interface. The binding pocket of CCR2-RA-[R] is highly enclosed and possesses a balanced combination of hydrophobic and polar features, all of which favors pocket “druggability”.
