|
|
| | 5-(TETRADECYLOXY)-2-FUROIC ACID Basic information |
| Product Name: | 5-(TETRADECYLOXY)-2-FUROIC ACID | | Synonyms: | TOFA;5-(tetradecyloxy)-2-furancarboxylicaci;5-tetradecyloxy-2-furonicacid;mdl14514;RMI-14514;TOFA (5-(Tetradecyloxy)-2-furoic acid);5-(TETRADECYLOXY)-2-FURANCARBOXYLIC ACID;5-(TETRADECYLOXY)-2-FUROIC ACID | | CAS: | 54857-86-2 | | MF: | C19H32O4 | | MW: | 324.45 | | EINECS: | | | Product Categories: | | | Mol File: | 54857-86-2.mol | 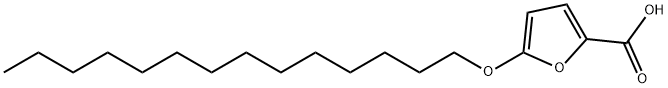 |
| | 5-(TETRADECYLOXY)-2-FUROIC ACID Chemical Properties |
| Melting point | 112-115 °C | | Boiling point | 441.7±25.0 °C(Predicted) | | density | 1.003±0.06 g/cm3(Predicted) | | storage temp. | -20°C | | solubility | DMSO: 2.5 mg/mL | | form | solid | | pka | 3.55±0.10(Predicted) | | color | white to beige | | Stability: | Stable for 1 year from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 1 month. |
| WGK Germany | 2 | | RTECS | LU0288000 | | HS Code | 29321900 |
| | 5-(TETRADECYLOXY)-2-FUROIC ACID Usage And Synthesis |
| Description | TOFA (54857-86-2) interferes with fatty acid synthase via inhibition of acetyl Co-A carboxylase (ACC1).1 Induces apoptosis in a variety of tumor cell lines.2,3 Stimulates neurite outgrowth and neuronal differentiation in rat pheochromocytoma cells.4 TOFA impairs glucose-stimulated insulin secretion after chronic treatment.5 | | Uses | TOFA has been used as a lipid biosynthesis inhibitor in mesenchymal stromal cells (MSCs), human pluripotent stem cells., an acetyl-CoA carboxylase 1 inhibitor in murine adipocyte cell lines., a lipolysis inhibitor in cancer stem cells (CSCs). | | Definition | ChEBI: A member of the class of furans that is 2-furoic acid in which the hydrogen at position 5 is replaced by a tetradecyloxy group. | | Biochem/physiol Actions | 5-(Tetradecyloxy)-2-furoic acid (TOFA) elicits hypolipidemic?functionality by favoring fatty acid breakdown and at the same time preventing biosynthesis. It induces apoptosis in pancreatic cancer cells and favors tumor suppression. | | References | References/Citations:
1) Halvorson?et al. (1984),?Inhibition of fatty acid synthesis in isolated adipocytes by 5-tetradecyloxy)-2-furoic acid; Lipids,?19?851
2) Guseva?et al. (2011),?TOFA (5-tetradecyl-oxy-2-furoic acid) reduces fatty acid synthesis, inhibits expression of AR, neuropilin-1 and Mcl-1 and kills prostate cancer cells independent of p53 status; Cancer Biol. Ther.,?12?80
3) Zhou?et al. (2003),?Fatty acid synthesis inhibition triggers apoptosis during S phase in human cancer cells; Cancer Res.,?63?7330
4) Schmidt?et al. (1999),?Transcription control and neuronal differentiation by agents that activate the LXR nuclear receptor family; Mol. Cell. Endocrinol.,?155?51
5) Ronnebaum?et al. (2008),?Chronic suppression of acetyl-CoA carboxylase 1 in beta-cells impairs insulin secretion via inhibition of glucose rather than lipid metabolism; J. Biol. Chem.,?283?14248 |
| | 5-(TETRADECYLOXY)-2-FUROIC ACID Preparation Products And Raw materials |
|