| Company Name: |
TargetMol Chemicals Inc.
|
| Tel: |
4008200310 |
| Email: |
marketing@tsbiochem.com |
| Products Intro: |
Product Name:Levallorphan;Levallorphan
CAS:152-02-3
Purity:98% Package:5 mg
|
|
| | levallorphan Basic information |
| Product Name: | levallorphan | | Synonyms: | 17-(2-propenyl)morphinan-3-ol;C07069;levallorphan;(-)-3-Hydroxy-N-allylmorphinan;17-Allylmorphinan-3-ol;2H-10,4a-Iminoethanophenanthren-6-ol, 11-allyl-1,3,4,9,10,10a-hexahydro- (6CI);l-N-Allyl-3-hydroxymorphinan;MCL 113 | | CAS: | 152-02-3 | | MF: | C19H25NO | | MW: | 283.41 | | EINECS: | 2057991 | | Product Categories: | | | Mol File: | 152-02-3.mol | 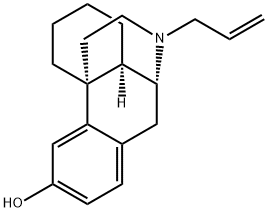 |
| | levallorphan Chemical Properties |
| Melting point | 180-182° | | alpha | D20 -88.9° (c = 3 in methanol) | | Boiling point | 425.96°C (rough estimate) | | density | 1.0260 (rough estimate) | | refractive index | 1.5000 (estimate) | | pka | 10.17±0.20(Predicted) |
| | levallorphan Usage And Synthesis |
| Definition | ChEBI: Levallorphan is a morphinane alkaloid. | | Brand name | Lorfan (Roche). | | Clinical Use | Levallorphan
is an opioid antagonist with practically
no analgesic action. It has been used as
one of the first relative pure antagonists for the
treatment of opioid overdosage, to reverse opioid
central depression and to antagonize opioidinduced
respiratory impairment . The compound
is now replaced by naloxone . | | Synthesis | Starting material for the levallorphan
synthesis is 1-(4-methoxybenzyl)-
1,2,3,4,5,6,7,8-octahydro isoquinoline (1)
.
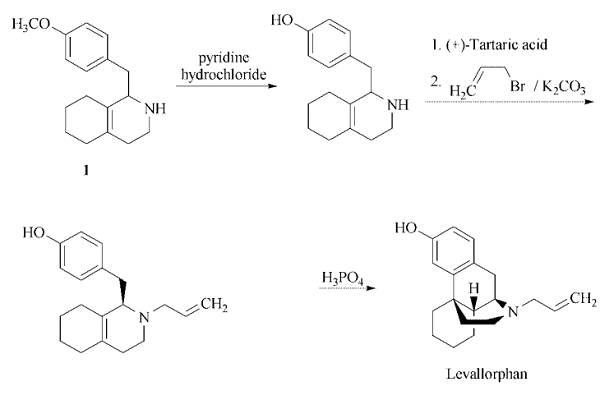 | | Purification Methods | It crystallises from aqueous EtOH. It is a narcotic antagonist. [Schneider & Grüssner Helv Chim Acta 34 2211 1951, Hellerbach Helv Chim Acta 39 429 1956.] |
| | levallorphan Preparation Products And Raw materials |
|