- AMMONIUM OLEATE
-

- $0.00 / 1kg
-
2024-07-02
- CAS:544-60-5
- Min. Order: 1kg
- Purity: 0.99
- Supply Ability: 50000kg
|
| | AMMONIUM OLEATE Basic information |
| Product Name: | AMMONIUM OLEATE | | Synonyms: | 9-Octadecenoicacid(Z)-,ammoniumsalt;9-octadecenoicacid,ammoniumsalt;oleicacid,ammoniumsalt;AMMONIUM OLEATE;9-Octadecenoic acid (9Z)-, ammonium salt;(Z)-9-Octadecenoic acid ammonium salt;aMMoniuMolate;ai3-36578 | | CAS: | 544-60-5 | | MF: | C18H37NO2 | | MW: | 299.49 | | EINECS: | 208-873-1 | | Product Categories: | | | Mol File: | 544-60-5.mol | 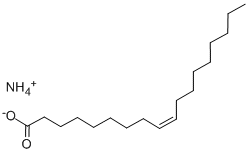 |
| | AMMONIUM OLEATE Chemical Properties |
| Melting point | 70-72°F | | Boiling point | 440.8°C (rough estimate) | | density | 0.9758 (rough estimate) | | refractive index | 1.5000 (estimate) | | solubility | soluble in H2O; slightly soluble in acetone | | form | yellow-brown paste | | color | yellow-brown paste | | Water Solubility | soluble H2O (27°C); slightly soluble acetone [MER06] | | LogP | 7.698 (est) | | EPA Substance Registry System | Ammonium oleate (544-60-5) |
| | AMMONIUM OLEATE Usage And Synthesis |
| Chemical Properties | Yellow to brownish ointmentlike mass; ammonia odor. Decomposes on heating. Soluble in water and hot alcohol. Combustible. | | Uses | Detergent, solidifying alcohol, demonstrating liquid crystals according to Lehmann. | | General Description | Yellow-brown paste with a weak odor of ammonia. Sinks and mixes with water. | | Air & Water Reactions | Water soluble. Gives basic solution | | Reactivity Profile | Salts, basic, such as AMMONIUM OLEATE, are generally soluble in water. The resulting solutions contain moderate concentrations of hydroxide ions and have pH's greater than 7.0. They react as bases to neutralize acids. These neutralizations generate heat, but less or far less than is generated by neutralization of the bases in reactivity group 10 (Bases) and the neutralization of amines. They usually do not react as either oxidizing agents or reducing agents but such behavior is not impossible. | | Hazard | Emulsifying agent, cosmetics. | | Health Hazard | Contact with eyes causes irritation. Prolonged contact may cause skin irritation. | | Fire Hazard | Special Hazards of Combustion Products: Toxic oxides of nitrogen may form in fire. | | Flammability and Explosibility | Not classified |
| | AMMONIUM OLEATE Preparation Products And Raw materials |
|