| Company Name: |
3B Pharmachem (Wuhan) International Co.,Ltd.
|
| Tel: |
821-50328103-801 18930552037 |
| Email: |
3bsc@sina.com |
| Products Intro: |
Product Name:17-PA;17-Phenyl-(3α,5α)-androst-16-en-3-ol
CAS:694438-95-4
Purity:99% HPLC Package:1Mg ; 5Mg;10Mg ;100Mg;250Mg ;500Mg ;1g;2.5g ;5g ;10g
|
| Company Name: |
EMMX Biotechnology LLC
|
| Tel: |
888-539-0666 |
| Email: |
info@emmx.com |
| Products Intro: |
Product Name:17-PA
CAS:694438-95-4
Purity:98% HPLC Package:5mg;10mg;25mg;50mg;100mg;250mg;500mg;1g
|
|
| Product Name: | 17-PA | | Synonyms: | 17-PA;17-PHENYL-(3A,5A)-ANDROST-16-EN-3-OL;17-Phenyl-(3α,5α)-androst-16-en-3-ol;Androst-16-en-3-ol, 17-phenyl-, (3α,5α)-;17-Phenyl-(3α,5α)-androst-16-en-3-ol;17PA,17 PA | | CAS: | 694438-95-4 | | MF: | C25H34O | | MW: | 350.54 | | EINECS: | | | Product Categories: | | | Mol File: | 694438-95-4.mol | 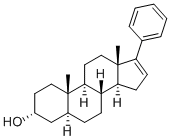 |
| | 17-PA Chemical Properties |
| storage temp. | Store at RT | | solubility | <8.76mg/ml in DMSO; <17.53mg/ml in ethanol |
| | 17-PA Usage And Synthesis |
| Uses | 17-PA is an antagonist of neurosteroid GABA potentiation. | | Biological Activity | Selective antagonist of neurosteroid potentiation and direct gating of GABA A receptors. Selectively reduces the effects of 5 α -reduced steroids compared to 5 β -reduced steroids and displays no effect on potentiation evoked by barbiturates and benzodiazepines. Attenuates 3 α ,5 α -THP-induced loss of righting reflex and total sleep time following i.c.v administration in rats. | | Enzyme inhibitor | This selective neurosteroid antagonist (FW = 350.54 g/mol; CAS 694438- 95-4; Soluble to 25 mM in DMSO and to 50 mM in ethanol), also known as 17-PA, targets neurosteroid potentiation and directly gates GABAA receptors. 17-PA inhibition was also useful in demonstrating that ethanol modulates the interaction of the endogenous neurosteroid allopregnanolone with the α1β2γ2L GABAA receptor. 17-PA selectively reduces the effects of 5α-reduced steroids compared to 5β-reduced steroids and displays no effect on potentiation evoked by barbiturates and benzodiazepines. It also attenuates 3α,5α-THP-induced loss of righting reflex and total sleep time, following intracerebroventricular administration in rats. |
| | 17-PA Preparation Products And Raw materials |
|